Detecting Microplastics in Foods and Beverages
Plastics and their degrading particles are polluting the world’s oceans and finding their way into the food supply.
Article Content
But plastic waste is not just limited to being an environmental contaminant and concern. There is now also growing evidence highlighting the effect anthropogenic activities are having on our global food chain.
To combat contamination in food, suppliers and producers regularly rely on robust and accurate characterization tools and methodologies to help identify the chemicals present. This helps them to uncover the source of the contamination and reduce the risk posed to humans. But the successful analysis of food and beverages requires detailed considerations to allow for clean and efficient analyses. Specifically, scientists need to consider how samples are prepared, and how the information can be used to gain insight into contamination.
Plastics: More Than Meets the Eye
Before considering the impact of plastics in food, it is important to outline the plastic problem in the environment. It is estimated that the world’s oceans contain about 270,000 tons of floating plastic waste (Ericksen et al. 2014). Much of the single-use plastics used in everyday activities are either non-biodegradable, or degrade slowly over the course of several decades.
However, this waste represents just one part of the plastic equation. When larger plastics (such as soda bottles, fishing nets, or plastic bags) degrade or decompose, they create smaller particles or fragments that can have a less visible impact on the environment. These smaller particles, which are less than 5 mm in length, are known as microplastics.
Microplastics can be broadly separated into two categories: primary and secondary. Primary microplastics refer to particles manufactured and added to commodities for specific purposes. Examples include microbeads that are often added to toothpastes and cosmetics to act as abrasives. These products eventually end up in waste water streams that culminate in the world’s oceans. Although these plastic particles are still commonly used in some products, regulations and legislation are being introduced in some countries to ban the use of primary microplastics. For instance, in 2015 the U.S. government passed a law prohibiting the manufacturing, packaging, and distribution of rinse-off cosmetics containing plastic microbeads, with other countries, including the UK, following suit in later years.
In contrast, secondary microplastics originate from the slow degradation of larger plastics. Common sources include plastic waste in landfills that degrades into smaller particles and permeates the water table before reaching the world’s oceans. Another unexpected source of microplastics is through washing clothes. Washing processes shed fibers that break down into plastic particles, which will end up in the world’s oceans.
Microplastics Enter the Food Chain
Regardless of whether microplastics are originally and purposely created by humans or not, it is in the world’s oceans where microplastics are suspected to be infiltrating the food chain. One mechanism for microplastic infiltration into the food chain is through zooplankton, which ingest microplastics (Cole et al. 2013). As zooplankton are at the bottom of the food chain, any microplastic contamination will be biomagnified up the food chain, ultimately reaching humans.
In recent years, multiple studies have shown that significant quantities of seafood consumed contained microplastic particles. In one study, researchers found that the average European consumer diet results in their exposure to approximately 11,000 microplastic particles (Van Cauwenberghe and Janssen 2014). More recently, research from the Medical University of Vienna, for the first time, demonstrated the presence of up to nine different types of microplastics in human feces (Schwabl et al. 2018). This study provided strong evidence that the origin of microplastics in humans was due to consuming contaminated food.
Several studies have been commissioned that have also confirmed the impact plastic waste is having on marine life. For instance, oyster reproduction was severely affected when exposed to polystyrene microparticles (Sussarellu et al. 2016). Although there is currently a limited understanding of the associated health risks of consuming microplastic contaminated food, it is known that microplastics accumulate various chemicals or bacteria throughout their lifetime. These contaminants can then enter the food chain with the microplastics and potentially cause consumer illness.
For instance, it is possible that pollutants in the water–such as polychlorinated biphenyls, polyaromatic hydrocarbons, heavy metals and pesticides–could adhere to the surface of microplastic particles. These are then “ingested” by marine life, become biomagnified up the food chain, before being exposed to consumers in the form of seafood. In addition, the manufacture of plastic packaging or primary microplastics can result in the introduction of chemicals that are harmful when consumed. For instance, polyvinyl chloride is generally loaded with plasticizers, which are known to be toxic to humans.
Identifying and Analyzing Plastics
Getting a handle on microplastics and what they mean for our planet, the food chain, and our bodies is becoming an increasing priority for environmental and food laboratories around the world. They require the right analytical research tools and best methodology practices to advance knowledge and ultimately spur new ideas and solutions.
Given the small size of microplastic particles, identification relies on using characterization methods. One widely used tool for analyzing microplastics is Fourier-transform infrared spectroscopy (FT-IR). This method analyzes the absorbance of energy by the chemical compounds present in a sample. The energies are then recorded to create a spectrum with the individual peaks corresponding to specific bonds in the chemical compound.
An FT-IR instrument can measure particles of various sizes, and an FT-IR infrared microscope is capable of identifying particles that are 250–5 µm (0.25–0.005 mm) in size, making it useful in microplastics analysis.
When specifically analyzing food, one of the most important factors to consider is the preparation of samples prior to analysis. Due to the widespread contamination of microplastics, a large variety of foods must be analyzed. For instance, microplastics could be present in drinking water, bottled water, sea salt, seafood, fish, and have even been found in non-marine animals, such as poultry fed using fish-based products.
The different varieties of food present a unique problem for analytical scientists: each sample matrix must be prepared to ensure that a clean and interpretable infrared spectrum is produced. For bottled water, sample preparation is relatively simple, with filtration isolating the microplastic particles that are then analyzed by the FT-IR microscope.
Food samples are more complicated to prepare. Instead of having a “clean” sample that is ready for filtration and analysis, marine or land animals require additional steps throughout preparation. One of the major considerations in food samples is the need to remove any organic or biological material that is present in order to isolate the microplastic particles before filtration and FT-IR analysis is performed. While removal of biological material is time-consuming, laboratories have standard protocols in place for the removal of these materials. Digestion is the most common approach, which uses acid, base, peroxide-based chemicals or even enzymes to break the materials apart and isolate the microplastic particles for filtration and analysis.
Bottled Water Analysis
One of the most recent studies investigating microplastics performed at PerkinElmer, Inc., Seer Green, UK, analyzed the microplastics present in five different anonymized samples of bottled water (Robertson 2018). The bottled water was filtered to obtain infrared-ready samples. Using a Spotlight 400 FT-IR Imaging system, these samples were then analyzed to identify the particles present.
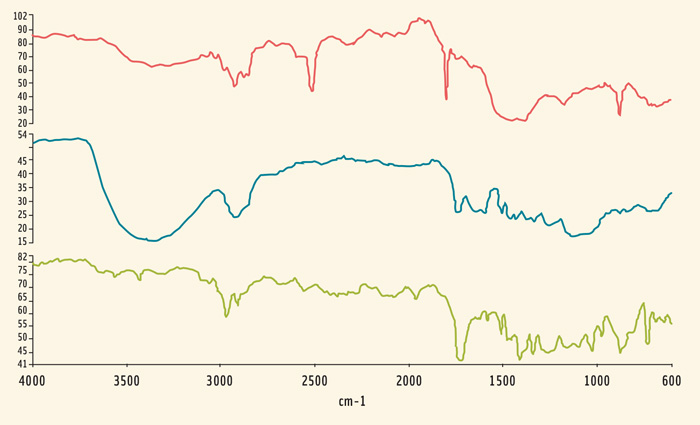
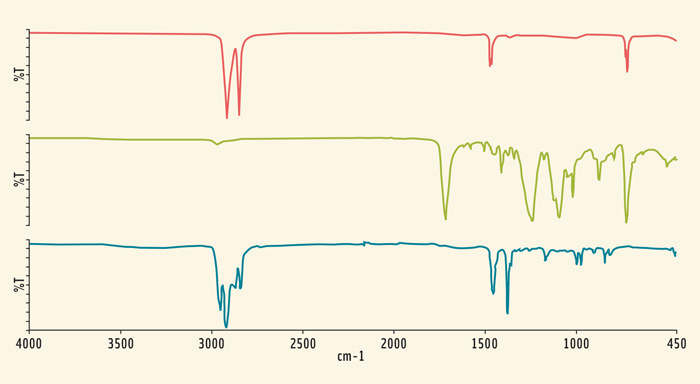
Every sample of bottled water was found to contain tens of plastic particles. The microplastic particles present varied in size of 200–20 µm (0.2–0.02 mm), with other fibers also identified that were larger than 2 mm in length. There was some commonality of the particle contamination between the bottled water samples. Characteristic chemical peaks in the chemical spectra helped to identify the compounds present. The peaks corresponded to the carbon backbones of the plastics polyethylene, polyethylene terephthalate (PET), and polypropylene. These spectra are shown in Figure 1. Importantly, these plastics are commonly used in the construction of plastic bottles themselves. Bottle lids are typically constructed from polyethylene, the bottle itself is typically PET, and finally the label can be made of various materials including polypropylene, coated paper, or other polymers with spectra shown in Figure 2.
The presence of these plastics in water samples has led to the suggestion that the manufacturing process could be the source of the contamination. However, additional chemical compounds were identified in the water samples. The majority of these were cellulose-based materials, including cellulose and cellulose acetate. In addition, other polymeric particles were also detected such as polyamide, polyvinyl alcohol, and c. In total, only around 5% of the particles identified from the water samples were polyethylene, polypropylene, or PET. The presence of these other chemicals indicates that contamination is not the sole result of manufacturing processes and instead occurs prior to production.
On the other hand, bottled water is often marketed as being a cleaner alternative to tap water. The study also tested this hypothesis, where bottled samples were compared with tap water from the laboratory in the UK. The two microscopy images are shown in Figure 3. These two images clearly show that tap water contains hundreds more fibers and plastic particles in the same sample volume than any of the bottled water samples. Interestingly, these were identified as the same particles as were present in the bottled water samples. This provides further evidence that the origin of microplastics in water is due to the widespread microplastic contamination of water.
Combating Future Contamination
 Plastic and microplastic contamination are harmful to both the environment and the food chain. While growing evidence indicates humanities increased exposure to microplastics, additional studies are needed that model the effects of microplastic contamination on consumer health. Identifying microplastics is just one step to combat contamination. Once the plastic chemical has been identified, the source needs to then be identified and addressed.
Plastic and microplastic contamination are harmful to both the environment and the food chain. While growing evidence indicates humanities increased exposure to microplastics, additional studies are needed that model the effects of microplastic contamination on consumer health. Identifying microplastics is just one step to combat contamination. Once the plastic chemical has been identified, the source needs to then be identified and addressed.
For identification and analysis purposes, FT-IR imaging equipment can be employed at various stages of food or bottled water manufacturing to provide information that helps reduce the risk to consumers. The microplastics community is now moving towards the use of standardized methods to analyze samples. This is essential to provide consistency at all stages of sample analysis, from preparation to measurement and reporting. It is only through the application of characterization instruments that products and commodities can be definitively identified as free from contamination and safe for consumption.
Ian Robertson is a senior applications scientist at PerkinElmer, Inc., Seer Green, UK ([email protected]).