New Intervention Processes for Minimally Processed Juices
Many processes are available to improve the safety of minimally processed fruits and vegetable juices
Continued outbreaks attributed to by pathogen-contaminated fresh juices has caused the regulatory agencies to rethink their position on how industry should implement a 5- log reduction.
 Previously, the Food and Drug Administration had suggested that the cumulative reduction delivered by external processes could be sufficient for industry to demonstrate a safe process for intact, sound fruit. Juices not receiving a 5-log reduction would be required to have a warning statement.
Previously, the Food and Drug Administration had suggested that the cumulative reduction delivered by external processes could be sufficient for industry to demonstrate a safe process for intact, sound fruit. Juices not receiving a 5-log reduction would be required to have a warning statement.
New evidence questions the assumption that the pathogens are located on the surface of the fruit, thus limiting the effectiveness of any surface treatment. Sapers et al. (1999) reported that Escherichia coli bacteria were bound or entrapped by cuts or punctures on apples, resulting in higher levels of contamination. Dye uptake by sound citrus fruit further supports the possibility that pathogenic bacteria may be internalized, limiting the affectiveness of surface treatments (Buchanan, 1999). Fortunately, several processes are available to provide safe, minimally processed juices.
 Limitations of Surface Treatments
Limitations of Surface Treatments
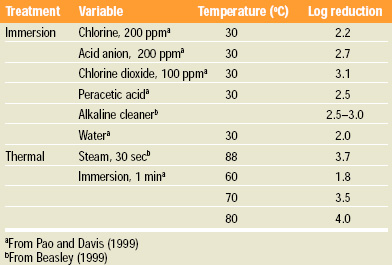
Chemical sanitizers, mechanical scrubbing, and thermal treatments have been shown to deliver significant reductions to surface-contaminated fruit (Table 1). Effective surface treatments can cumulatively contribute as a reduction process to the extent that the contamination is distributed on the surface of the fruit. For example, if 99% of the natural contamination is on the surface and 1% is internal, then only a 2-log reduction can be realized even if the surface is completely sterilized!
To date, very little is known about the distribution of naturally occurring pathogens on fruit. When Pao and Davis (1999) demonstrated an overall 5-log kill of E. coli on the surface of oranges, the resulting juice was reduced by only 3.5 logs, compared to the untreated surface-contaminated fruit. In the same study, the natural microbiological load of the orange juice was reduced by only 1.5–2.0 logs, despite the application of >5-log surface treatment. This indirect evidence supports the hypothesis that natural contamination is distributed throughout the fruit. Thus, active intervention processes must be used on the juice when surface sterilization is inadequate to accomplish the recommended 5-log reduction.
Despite the numerous outbreaks attributed to fresh juice, very few events have been linked to pack-house fruit. Either the consumer recognizes contaminated, spoiled fruit and does not eat it, or the event is so infrequent that it is not linked to the fruit. It is also conceivable that in a fresh-juice plant, a single infected fruit could contaminate an entire tank of juice, resulting in a widespread out-break. Conversely, in-store juice extractors that have small-volume reservoirs would contaminate very few containers if a single contaminated fruit were extracted.
Minimal Processes for Juice
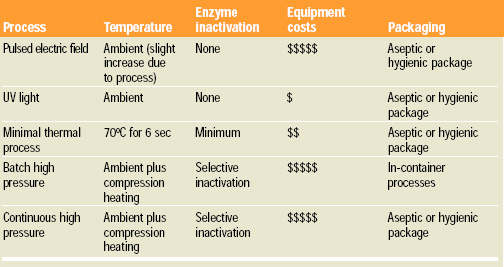
Several effective new process technologies are available to accomplish a microbiological reduction in juices. Pulsed electric fields, ultraviolet light, minimal thermal processes, and batch and continuous high-pressure processing systems have been offered commercially. The applicability of each technology to a specific juice depends on the characteristics of the product and the pathogens of interest which may be resistant to the process. Each of the minimal processes is intended for a reduction of pathogens and does not accomplish a kill adequate for commercial sterility. As such, products must be maintained under refrigerated storage and distribution to slow spoilage.
--- PAGE BREAK ---
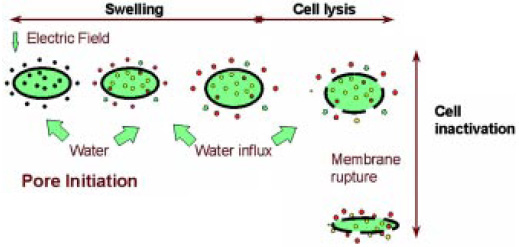 • Pulsed Electric Fields. PEF processing subjects the juice to a high-voltage electric field on the order of 25,000–70,000 V/cm for a few microseconds. The intense field electroporates the microorganisms, resulting in loss of membrane integrity, inactivation of proteins associated with channels, and leakage of cellular contents (Jeyamkondan et al., 1999) (Fig. 1). The application of many rapid high-power pulses can significantly increase the juice temperature unless this generated heat is removed using external cooling units.
• Pulsed Electric Fields. PEF processing subjects the juice to a high-voltage electric field on the order of 25,000–70,000 V/cm for a few microseconds. The intense field electroporates the microorganisms, resulting in loss of membrane integrity, inactivation of proteins associated with channels, and leakage of cellular contents (Jeyamkondan et al., 1999) (Fig. 1). The application of many rapid high-power pulses can significantly increase the juice temperature unless this generated heat is removed using external cooling units.
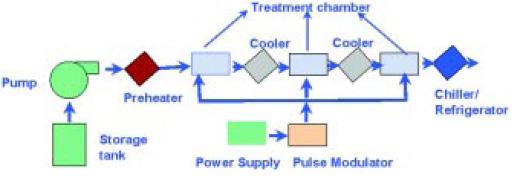 PEF flow systems typically consist of multiple treatment cells connected by stainless-steel tubing in series (Fig. 2). The juice is conveyed in sequence to the process cells and receives 20–100 pulses cumulatively. Uniformity of the treatment is accomplished using turbulent flow and through geometric design of the treatment chamber. Fruit pulp or air bubbles will distort the electric field, resulting in process nonuniformity and bacterial survival.
PEF flow systems typically consist of multiple treatment cells connected by stainless-steel tubing in series (Fig. 2). The juice is conveyed in sequence to the process cells and receives 20–100 pulses cumulatively. Uniformity of the treatment is accomplished using turbulent flow and through geometric design of the treatment chamber. Fruit pulp or air bubbles will distort the electric field, resulting in process nonuniformity and bacterial survival.
Products are minimally affected by the process. Since damage occurs on a cellular level, flavor and enzyme activity are not significantly diminished. Consumer perception of PEF-treated products depends on how long the product has been stored under refrigeration after processing. Product deterioration occurs as a result of the enzyme activity and naturally occurring chemical changes in the product.
 • Ultraviolet Light. Microbial inactivation of microbes with UV light (Fig. 3) is a well-known surface treatment. Inactivation is caused by the cross-linking of thymine dimers of the cells’ DNA, thus preventing repair and reproduction. The germicidal wavelength for inactivation is in the range of 240–265 nm. Continuous 254-nm lights using low- and medium-pressure UV lights have historically been the source of choice, although interest in excimer lamps and lasers at 247 nm is increasing. Microbial populations in the range of 10/cm may lead to shadowing and may hinder inactivation.
• Ultraviolet Light. Microbial inactivation of microbes with UV light (Fig. 3) is a well-known surface treatment. Inactivation is caused by the cross-linking of thymine dimers of the cells’ DNA, thus preventing repair and reproduction. The germicidal wavelength for inactivation is in the range of 240–265 nm. Continuous 254-nm lights using low- and medium-pressure UV lights have historically been the source of choice, although interest in excimer lamps and lasers at 247 nm is increasing. Microbial populations in the range of 10/cm may lead to shadowing and may hinder inactivation.
Historically, UV processing has been limited to surface disinfection because of the poor penetration and shadowing effects. A petition was filed recently by California Day-Fresh Foods and Salcor Engineering to allow use of UV light for pasteurization of juices. The technology breakthrough that led to this use of UV light was the design of treatment chambers utilizing turbulent flow to form a “continuously renewed surface,” thus enabling microbial inactivation to be achieved. Typical penetration of UV light in juices is on the order of 1 mm for absorption of 90% of the light. By designing the treatment chamber using transparent Teflon™ tubing wound in a helix, the designers assured turbulent flow with secondary effects due to the Dean effect. This provided the continuously renewed surface necessary for microbial inactivation in juices.
In a food plant environment, UV processing must be carefully controlled to achieve the desired microbiological reduction. Flow rate, absorption, particle size, and UV light intensity are among the critical control points for the process. Extensive experience in UV processing of water indicates that UV systems will provide many hours of satisfactory performance.
• Minimal Thermal Processes. If thermal processing of juices is one of the oldest and safest methods of food preservation, why is it being reinvented today as a minimal process? The answer lies in the consumer’s desire to have a juice which has the appearance and taste of a freshly squeezed product. The minimum safe treatment of high-acid products is about 70ºC for 6 sec. For many juices, this treatment assures a 5-log reduction of acid-tolerant E. coli O157:H7 but does not inactivate spoilage organisms or affect deteriorative enzymes. The resulting product must be kept refrigerated and has the characteristics of a fresh product.
--- PAGE BREAK ---
 Very few juice products require minimal thermal processes to maintain sensitive flavor characteristics. Most of the fruit juices, such as orange, grapefruit, cranberry, grape, and apple juice, are sufficiently robust to tolerate thermal processes in the range of 90–100ºC. However, vegetable juices such as carrot or spinach may have flavor or color changes at elevated temperatures. For these products, minimal thermal or nonthermal processes may be necessary.
Very few juice products require minimal thermal processes to maintain sensitive flavor characteristics. Most of the fruit juices, such as orange, grapefruit, cranberry, grape, and apple juice, are sufficiently robust to tolerate thermal processes in the range of 90–100ºC. However, vegetable juices such as carrot or spinach may have flavor or color changes at elevated temperatures. For these products, minimal thermal or nonthermal processes may be necessary.
• Batch High-Pressure Processing. HPP dates back to the late 19th century when Hite (1899) used pressure to inactivate microorganisms. Although the principle is unchanged, the technology to accomplish HPP on a commercial scale is now beginning to be available. Process treatments of 500-700 MPa (75–105,000 psi) for 2 min at an initial temperature of 25ºC have been shown to be adequate to inactivate most pathogens of interest in fruit juices.
 Juices to be processed using batch systems (Fig. 4) are first placed in containers which can survive the intensive process. The containers are then placed into a crate and loaded into a pressure vessel for processing. The overall process requires about 5 min, with loading requiring 1 min, pressurization 1 min, processing 2 min, and depressurization and unloading 1 min. In practice, the process resembles a conventional batch retort operation but at a much lower temperature.
Juices to be processed using batch systems (Fig. 4) are first placed in containers which can survive the intensive process. The containers are then placed into a crate and loaded into a pressure vessel for processing. The overall process requires about 5 min, with loading requiring 1 min, pressurization 1 min, processing 2 min, and depressurization and unloading 1 min. In practice, the process resembles a conventional batch retort operation but at a much lower temperature.
High-pressure-processed juices retain a fresh-like flavor and appearance. Inactivation of pectin methylesterase in orange juice has been shown to be difficult, resulting in some product instability and settling. High-pressure-treated clear juices such as apple, pear, and white grape juice are remarkably similar to fresh product.
Bacterial inactivation has recently been modeled using conventional kinetics with additional variables to account for the accelerated destruction at high pressure (Roviere et al., 1996). Mussa et al. (1999) determined that for Listeria monocytogenes, an increase in pressure of 167 MPa was equivalent to raising the temperature of the process by 10ºC. For L. monocytogenes, a typical HPP process at 500 MPa could be done at a 30ºC lower temperature than the corresponding thermal treatment. Compression heating of the product attributed to the adiabatic temperature rise has been shown to result in a composition-dependent 2–4ºC increase per 100 MPa of process pressure. This temperature increase is proportional to the pressure rise and drops instantaneously as the pressure is released. The combined compression heating and kinetic pressure effects contribute to the lethality of the process.
![]() • Continuous High-Pressure Processing. Flow International Corp. developed semicontinuous HPP systems that can operate continuously as part of an aseptic system (Fig. 5). The product is pumped directly into the pressure vessel for processing and is separated from the pressurizing media by an isolator. Pressure is transmitted from the pressurizing liquid across the isolator to the product. After processing, the pressure is released and the product is pumped to a sterile tank. To minimize the chance of contamination after the process, sterile water is used as the pressurizing and transfer media.
• Continuous High-Pressure Processing. Flow International Corp. developed semicontinuous HPP systems that can operate continuously as part of an aseptic system (Fig. 5). The product is pumped directly into the pressure vessel for processing and is separated from the pressurizing media by an isolator. Pressure is transmitted from the pressurizing liquid across the isolator to the product. After processing, the pressure is released and the product is pumped to a sterile tank. To minimize the chance of contamination after the process, sterile water is used as the pressurizing and transfer media.
--- PAGE BREAK ---
The advantages of a semicontinuous system include rapid filling and emptying and the flexibility to use containers which cannot withstand the high pressure. Cups, brick, or gable-top cartons associated with aseptic packaging are readily adapted to semicontinuous HPP.
Integration of the HPP system into the aseptic system requires significant changes in the line sterilization process, since the pressure vessels and gaskets cannot withstand conventional steam sterilization. Combinations of pressure, temperature, and sanitizers may be utilized to accomplish presterilization of the system. A typical HPP aseptic line would utilize steam to sterilize product lines, aseptic tank, and aseptic packaging fill system. The pressure chamber, isolator, and valves on the HPP system would be sterilized by pressure, leaving a short distance from the outlet valve to the product line to be sterilized chemically.
Validation Tests Needed
The decision that fresh juice processors face today is whether alert consumers that unpasteurized juices can contain bacteria that pose a special risk to certain individuals, or to implement an intervention process which will truly improve the safety. While external treatments of fruit can demonstrate a 5-log reduction in surface contamination, the safety of freshly squeezed juices will not be assured. Until the distribution of the naturally occurring pathogens can be determined, it will be necessary to validate a 5-log reduction in the juice. Many satisfactory minimal processes are available which will improve product safety.
Since many of the minimal processes described above are new intervention processes, it is important that processors conduct appropriate microbiological validation tests. Processors further need to address all of the appropriate regulations, including the requirement for premarket approval (Larkin and Spinak, 1997; Hansen, 1999).
The decision that fresh juice processors face today is whether to alert consumers...or to implement an intervention processs which will truly improve the safety.
C.E. SIZER AND V.M. BALASUBRAMANIAM
The authors, Professional Members of IFT, are, respectively, Professor and Director, and Associate Professor of Food Process Engineering, National Center for Food Safety and Technology, Illinois Institute of Technology, Moffett Campus, 6502 S. Archer Road, Summit-Argo, IL 60501. Send reprint requests to author Sizer.
Edited by Neil H. Mermelstein
Senior Editor
References
Beasley, L. 1999. Treatment options for fresh-squeezed citrus juices. Presented at NCFST/IFT Ann. Symp., “Minimally Processed Fruit and Vegetable Juices: Scientific Issues to Achieve a Safe Product,” Chicago, May 12-13.
Buchanan, R. 1999. Dye uptake study. Presented at public meeting with American Fresh Juice Council, Aug. 25.
Hansen, P.A. 1999. Routes to regulatory clearance for new intervention processes. Presented at NCFST/IFT Ann. Symp., “Minimally Processed Fruit and Vegetable Juices: Scientific Issues to Achieve a Safe Product,” Chicago, May 12-13.
Hite, B.H. 1899. The effect of pressure in preservation of milk. Bull. 85, p. 15. West Virginia Univ. Agr. Exp. Sta., Morgantown. Cited in Hoover, D.G., Metrick, C., Papineau, A.M., Farkas, D.F., and Knorr, D. 1989. Biological effects of high hydrostatic pressure on food microorganisms. Food Technol. 43(3): 99-107.
Jeyamkondan, S., Jayas, D.S., and Holley, R.A. 1999. Pulsed electric field processing of foods: A review. J. Food Protect. 62: 1088-1096.
Larkin, J.W. and Spinak, S.W. 1997. Aspects of new technology implementation in the U.S. Presented at 1997 New Technologies Symp., Natl. Food Processors Assn. Convention, Chicago, Oct. 29.
Mussa, D.M., Ramaswamy, H.S., and Smith, J.P. 1999. High-pressure destruction kinetics of Listeria monocytogenes on pork. J. Food Protect. 62: 40–45.
Pao, S. and Davis, C.L. 1999. Enhancing microbiological safety of fresh orange juice by fruit immersion in hot water and chemical sanitizers. J. Food Protect. 62: 756-760.
Roviere, P., Moggi, A., Scaramuzza, N., Gola, S., Miglioli, L., Carpi, G., and Dall’Aglio, G. 1996. High-pressure heat treatments: Evaluation of the sterilizing effect and other thermal damage. Industria Conserve 71: 473-483.
Sapers, G.M., Miller, R.L., and Mattrazzo, A.M. 1999. Effectiveness of sanitizing agents in inactivating Escherichia coli in golden delicious apples. J. Food Sci. 64: 734-737.
Tsong, T.Y. 1990. Review on electroporation of cell membranes and some related phenomena. Biochem. Bioeng. 24: 271-295.
