Kinder, Gentler Food Processing


Food is processed to meet targeted sensory attributes and minimum shelf life and to eliminate the risk of disease transmission by the processed product. Processors traditionally used heat to meet these goals. Heat can reliably inactivate spoilage and pathogenic organisms and enzymes that contribute to quality deterioration. Contrary to these benefits, excessive thermal processing negatively impacts the nutritional and sensorial qualities of food. Alternatives to heat have been implemented to overcome these drawbacks, but these newer processes lag in effectiveness against some spoilage and pathogenic agents, particularly bacterial spores, in comparison to heat. In recent decades, thermal and nonthermal treatments are combined to produce food that meets the processing goals just described. The success of this approach is well-exemplified by the emergence of the “extended shelf life (ESL) food” category. In this article, we will discuss processes that make ESL products possible and address ways to make these foods safe through the use of selected emerging processing technologies, particularly high pressure. To simplify this discussion, initially we will consider the thermal approach to processing ESL food. Additionally, special attention will be given to high-moisture low-acid foods. The following is a brief account of technologies that border ESL processes—i.e., pasteurization and sterilization.
Commercial Sterilization
Thermal sterilization involves heat-treating food at temperatures greater than 100°C, hence steam pressure is applied during this thermal treatment. The temperature-time combination should be sufficient to inactivate the most heat-resistant bacterial spores of concern in the product. Because of their high heat resistance, Clostridium botulinum spores are considered the cause of safety concern in high-moisture low-acid commercially sterilized food, hence sterilization processes are designed to eliminate these spores. The process similarly eliminates spores of other pathogens likely found in products prior to processing. Products that receive commercial sterilization treatment are microbiologically stable at normal storage temperature and can have shelf life up to three years. Harsh thermal processes involved in sterilization degrade heat-sensitive nutrients and may damage product quality.
Thermal Pasteurization
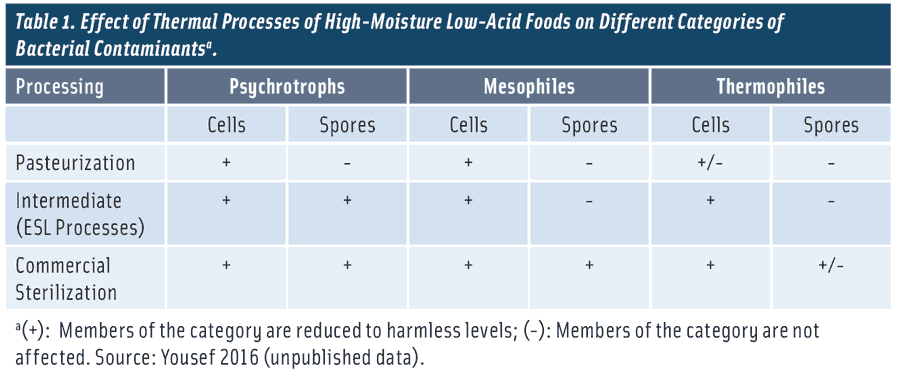
This technology involves application of mild heat with considerable lethality to vegetative cells of psychrotrophic and mesophilic bacteria, whether these are spoilage or pathogenic species (Table 1). The heat applied is often between 55°C and 80°C for a holding time that varies with the applied temperature. Cells of thermophilic bacteria may not be inactivated by pasteurization, and the process does not inactivate bacterial spores or heat-resistant fungal spores. The process parameters (temperature and time) are selected based on the thermal susceptibility of the cells of the most heat-resistant pathogen of concern in the treated product. Acid foods (pH < 4.6) can be pasteurized at relatively mild thermal treatment; product acidity sensitizes microorganisms to heat (Silva and Gibbs 2010). Refrigeration of pasteurized food is essential to prevent the transformation of surviving spores into vegetative cells that can spoil the product or render it unsafe for human consumption. Considering that pasteurization doesn’t kill spores of psychrotrophic bacteria, the treatment may activate these spores and increase their propensity to germination. Trans-formation of these spores into vegetative cells that could multiply to high population would eventually spoil the pasteurized product, thus terminating its shelf life. If these psychrotrophic spore-formers are pathogenic species, their germination and outgrowth in pasteurization products would be a potential hazard.
Extended Shelf Life Foods
Although shelf life of a sterilized food can be measured in years, it is only a few days or weeks for pasteurized products. This short shelf life creates inconveniences to processors of pasteurized food. Pasteurized products should be quickly distributed to their destination market and disposed of properly if not sold by their short sell-by date. This increases food waste and drives up costs for the manufacturer. Hence, the ESL category has emerged as products receiving processes that are intermediate between pasteurization and sterilization, with shelf life expected to be longer than that of the pasteurized counterpart. An ESL thermal process should be sufficient to eliminate the microorganisms responsible for the termination of the shelf life of pasteurized products, i.e., spores of psychrotrophic bacteria (Marth 1998). This may be achieved at temperatures greater than 80°C and less than 100°C. ESL processes that inactivate spores of psychrotrophic bacteria would also inactivate degradative enzymes that may survive pasteurization. A typical thermal ESL process involves heating food at 90°C for 10 minutes (FSA 2008, Silva and Gibbs 2010). Considering that these processes do not eliminate other categories of bacterial spores, foods receiving ESL processes should be held refrigerated.
To reduce ESL process severity and further increase product shelf life, food processors often employ multiple hurdle technologies (Silva and Gibbs 2010). This includes the use of combinations of heat and natural antimicrobial agents. ESL processed low-acid foods may also be packaged under a modified or controlled-atmosphere gaseous environment. Use of gases such as CO2 and N2 to modify headspace atmosphere of packaged foods can help to retard deteriorative reactions and extend the shelf life (Brody 2003). It is worth noting that modified atmosphere packaging by itself is not lethal to harmful microorganisms. In fact, modified atmosphere may selectively encourage growth of anaerobic pathogens. Other hurdle technologies may involve the use of active and “intelligent” packaging systems.
Although thermal processing is effective in inactivating harmful pathogens and reducing food spoilage organisms, maintaining the cold chain is critical for ensuring microbial safety and shelf life of ESL low-acid foods. Potential temperature abuse (deviation from refrigeration temperature) during storage, distribution, or at the retail stores may promote germination of surviving spores. Production of botulinum neurotoxin due to the proliferation of non-proteolytic C. botulinum is the main concern for the safety of these in-pack processed, extended shelf life refrigerated foods (Peck and Stringer 2005). Psychrotrophic clostridia, including non-proteolytic C. botulinum, have been problematic in vacuum packaged beef, pork, and lamb; pasteurized crabmeat; and cooked turkey meat. While sodium nitrite is used to control C. botulinum in certain seafood and meat products, increasing consumer demand for clean label chemical preservative–free food products necessitates its removal. The demand for clean label foods also drives the need for development and validation of new generation processing and natural ingredient technologies.
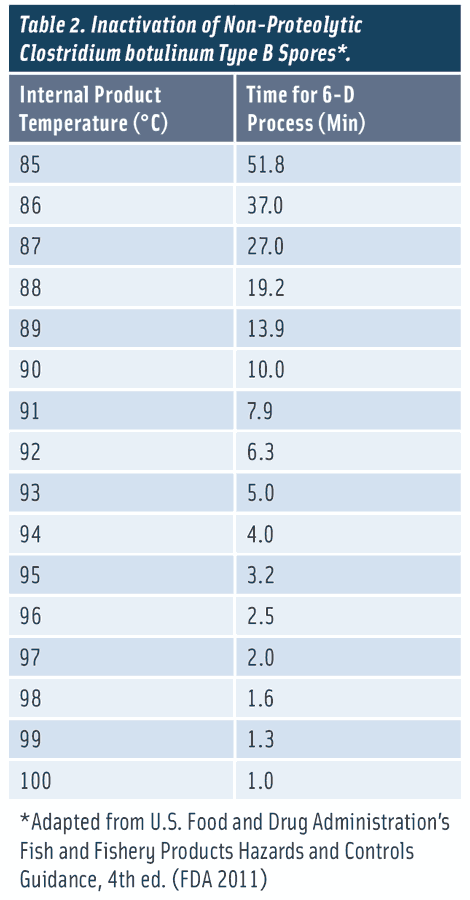
The U.S. Food and Drug Admini-stration (FDA) Fish and Fishery Products Hazards and Controls Guidance document identified C. botulinum type E and non-proteolytic types B and F as pathogens of concern for low-acid ESL foods packaged in a reduced- oxygen (e.g., vacuum packaged or modified atmosphere packaged) environment and stored refrigerated (FDA 2011). A process that delivers a 6-log reduction of non-proteolytic C. botulinum spores has been generally accepted by the FDA and European regulators, as well as the scientific community and food industry for ensuring the safety of chilled foods with extended shelf life (FDA 2011). Table 2 provides examples of 6-D thermal processes considering the most heat-resistant non-proteolytic C. botulinum spores (type B) as the target pathogen (FDA 2011).
Emphasis on Minimal Processing and Clean Label Products
Historically, consumer shopping decisions for food are primarily influenced by taste, price, and convenience. Thermal processing has been the technology of choice for preserving foods over the last 200 years. Recently, consumers have been placing emphasis on health and wellness, safety, and environmentally friendly processes. This led to increased demand for minimally processed foods, particularly natural and organic foods (Anon. 2016). In addition to quenching thirst, consumers seek beverages that deliver healthy nutrients and bioactive compounds. Health-conscious consumers also pay close attention to the ingredients in processed foods. They often read the label statement with the mindset of “can the ingredients on the label be found in my own kitchen?” as means to decide whether or not to purchase a product. They tend to avoid products perceived to contain artificial or chemical-sounding ingredients, although such ingredients may be scientifically proven safe and functional. This led the food industry to put more emphasis on clean label–oriented process and product development. A variety of factors can help the processor to achieve “clean labels.” This includes absence or minimal use of artificial additives, colors, preservatives, flavoring agents, and sweeteners. Currently, the clean label market is estimated at $62 billion in the United States and $165 billion globally (Nunes 2016). Processors often need to identify answers to the following questions as they consider introducing novel processing methods (West 2016):
1) Can the processed food maintain natural fresh taste? Alternatively, can it be formulated to maintain the “home-prepared” appeal?
2) Can the product be formulated to have cleaner labels without reducing shelf life or elevating food safety risk?
3) Is it possible to reduce the food waste as low as possible during handling, distribution, and at the retail level?
--- PAGE BREAK ---
Nonthermal Processing
With increased consumer demand for minimally processed foods, the food industry is embracing the use of nonthermal lethal agents such as high pressure, irradiation, pulsed electric field, and ultraviolet (UV) treatment (Zhang et al. 2011). Among the nonthermal technologies, food irradiation has been one of the most extensively studied food processing technologies since the 1960s, yet it is still considered a relatively “new” technology with limited market share. Commercial application of ionizing radiation to foods in the United State has grown slowly. Consumers’ lack of awareness of potential technology benefits has been a major hurdle for widespread acceptance of gamma radiation. Coordinated education efforts may help to increase consumer awareness of the beneficial aspects of such technologies (Eustice and Bruhn 2013).
The National Advisory Committee on Microbiological Criteria for Foods (NACMCF) expanded the definition of pasteurization to include both advanced thermal (ohmic, microwave heating) as well as nonthermal lethal (high pressure, UV radiation, pulsed electric field) agents as a part of processes leading to pasteurization (NACMCF 2006). Accordingly, pasteurization is redefined as “any process, treatment, or combination thereof, that is applied to food to reduce the most resistant microorganism(s) of public health significance to a level that is not likely to present a public health risk under normal conditions of distribution and storage.”
Markland et al. (2013) particularly recommended a high level of vigilance of Bacillus cereus, B. weihenstephanensis, and C. botulinum type E spores owing to their inherent resistance to processing and the innate ability of their vegetative cells to conduct metabolic activities at refrigeration temperatures. Although nonthermal processing methods are effective in reducing non-spore-forming bacterial populations, with the exception of gamma radiation, these methods do not inactivate bacterial spores. Thus, careful attention must be paid to maintain a proper cold chain during nonthermally pasteurized low-acid product handling, storage, and distribution.
The mechanism of action of nonthermal lethal agents on microorganisms and enzymes may be different from that of thermal processing. Unlike thermal processing, nonthermal methods have shown variable efficacy against enzymes. Hence, both thermal and nonthermal lethal agents are combined to achieve key food processing objectives, including enzyme inactivation. It is important to test the effect of nonthermal lethal agents with or without heat on a wide variety of microorganisms, enzymes, and nutrients.
High Pressure Processing
High pressure processing (HPP) is essentially a batch process, wherein the prepackaged food is suspended within a pressure chamber containing water and is subjected to pressure treatment for a specific duration. Unlike heat, pressure can be rapidly applied throughout the sample volume, regardless of sample size. Pressure treatment is effective against vegetative bacteria, but bacterial spores are not inactivated. Thus, it is important to store pressure-treated products under refrigerated temperature conditions.
 Typically, products are pressure-treated at chilled or ambient temperature conditions. The microbiological shelf life and quality of foods can be substantially extended by the use of HPP (Balasubramaniam et al. 2008). With the commercial introduction of pressure-treated guacamole by 1997, high pressure processing has been embraced by U.S. food processors to pasteurize a variety of value-added products, including seafood, meat, salsa, salad, juices, and fruit and vegetable products (Farkas and Hoover 2000).
Typically, products are pressure-treated at chilled or ambient temperature conditions. The microbiological shelf life and quality of foods can be substantially extended by the use of HPP (Balasubramaniam et al. 2008). With the commercial introduction of pressure-treated guacamole by 1997, high pressure processing has been embraced by U.S. food processors to pasteurize a variety of value-added products, including seafood, meat, salsa, salad, juices, and fruit and vegetable products (Farkas and Hoover 2000).
Though originally introduced as a post-packaging pasteurization technology, like heat, pressure can be used to modify various food constituents, including proteins and carbohydrates. This provides various product development opportunities for food processors, including creation of novel food textures, alteration of water binding capacity and rheological characteristics, and development of pressure-induced gel structures. Pressure treatment at chilled or moderate temperatures can preserve bioactivity and various food quality attributes such as flavor, color, and nutritional properties (Balasubramaniam et al. 2008). This enables the food processors to utilize both heat and pressure to create novel food products with consumer-desired quality attributes and extended shelf life.
Like thermal processing, various formulation and preprocessing steps can play a role in deciding final sensorial characteristics and shelf life of pressure-treated foods. For example, pressure treatment is often used as post-lethality treatment in deli meats, wherein the sealed package of precooked meat is pressure treated. The meat may be formulated with natural antimicrobial agents. Such a hurdle approach reduces or eliminates pathogen levels resulting from post-lethality contamination. Similarly, pasta products may be cooked prior to pressure pasteurization treatment to improve consumer sensorial experience. On the other hand, freshly extracted raw fruit juice or a formulated tomato salsa product may be pressure processed to ensure its microbiological safety without compromising fresh-like quality attributes. Due to their high acid content, pressure pasteurized acidic products are microbiologically stable. However, such products are often distributed under refrigerated conditions to preserve product quality during extended shelf-life.
Processing Combination to Achieve ESL
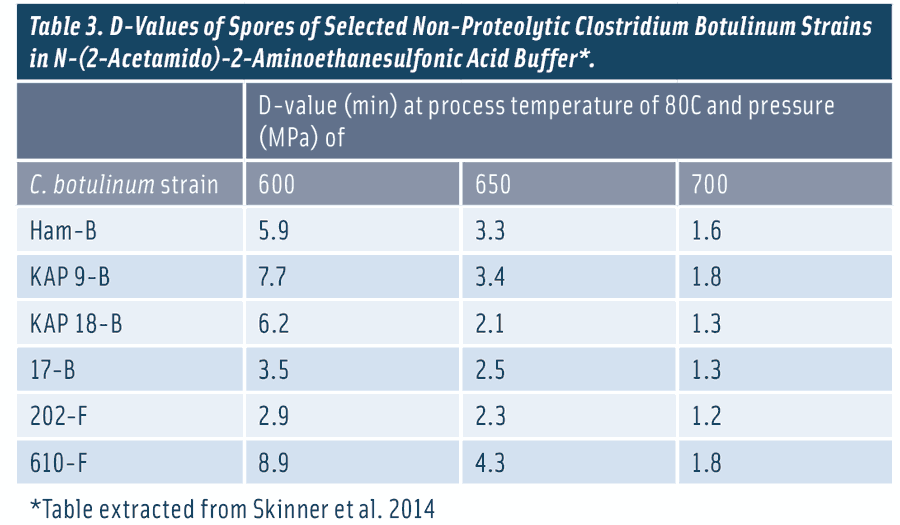
Researchers have been evaluating the efficacy of pressure-thermal combinations for production of ESL products. Legan et al. (2008) reported that the log reductions of non-proteolytic C. botulinum spores subjected to the combined high pressure and heat treatments were much greater than those subjected to the thermal treatment only. Daryaei et al. (2013) reported that a 600-MPa-thermal (60oC–85oC) treatment inactivated B. cereus spores suspended in cooked rice by 2.2–3.4 log during the 30-s pressure come-up time and to below the detection limit (> 7 logs) after 4- to 8-min pressure-holding times. In contrast, a 180-min treatment time was required to inactivate B. cereus spores to an undetectable level at atmospheric pressure (0.1 MPa) and 85oC. More recently, Skinner et al. (2014) screened multiple strains of non-proteolytic C. botulinum spores suspended in buffer (pH 7) for resistance to combined pressure (600, 650, and 700 MPa) and thermal (80°C) treatment (Table 3). The researchers identified the most resistant strains of the non-proteolytic C. botulinum spores to the combined pressure and thermal treatment and proposed using these strains for validation studies for ESL foods.
Pressure-thermal processes for production of extended shelf-life foods can provide a variety of benefits beyond food safety and shelf-life extension. For example, it has been reported that pressure treatment of foods may provide desirable sensory attributes at lower salt, polyphosphates, or emulsifier levels (Rodrigues et al. 2015, Martínez-Monteagudo et al. 2017). Reduced thermal exposure during ESL treatment can help to preserve heat-sensitive bioactive compounds. In comparison to thermal treatment (75oC–105oC, 0.1 MPa), combined pressure-thermal treatment (75oC–105oC, 600 MPa), reduced the formation of furan in liquid juices. More systematic process design and development studies are necessary to understand the synergistic, additive, or antagonistic effects of pressure and heat on microorganisms, food quality attributes, and nutrients.
 Where the ESL Technology Is Headed
Where the ESL Technology Is Headed
Currently in the United States, there are no standard definitions or regulatory requirements for ESL foods, including those produced by combined pressure-thermal treatment; no such product exists commercially in the United States. However, it is encouraging that well-studied treatment combinations can receive regulatory approval. For example, the FDA had no objections to two industrial petitions for producing shelf-stable low-acid foods by pressure-assisted thermal processing (IFSH 2015, Stewart et al. 2016). The recently implemented FDA Food Safety Modernization Act (FSMA) regulations require that processors employ risk-based procedures, practices, and processes to significantly minimize or prevent all known or reasonably foreseeable food safety hazards that require a preventive control. Selection of the target pathogen of concern is critical to process validation and verification for ensuring the microbiological safety of foods (Federal Register 2015). Considering these developments, current status of knowledge, and the market need, it is possible to produce successfully safe and wholesome ESL products for new generation consumers. However, this move should be accompanied with systematic studies addressing potential safety, quality, and technology challenges associated with the production of ESL food.
--- PAGE BREAK ---
 Concluding Remarks
Concluding Remarks
Although there is no regulatory definition of ESL, the ESL term applies to food developed by various technological solutions for enhancing its shelf life under refrigerated storage without compromising microbiological safety of the processed product. ESL products enable food processors to manufacture wholesome, nutritious foods with consumer-desired quality attributes. Thermal processing has been the technology of choice for ESL foods. In addition to heat, food scientists and engineers have a variety of nonthermal lethal agents (including high pressure, electric field, cold plasma, UV, gases, and natural antimicrobials) in their tool box for processes designed to produce clean label ESL foods. Such treatments also help to reduce thermal impact on heat-sensitive nutrients and food quality. Among these lethal agents, high pressure–based processing is leading the way in many food applications, including the opportunity to formulate novel food structures. To fully utilize the potential of nonthermal and thermal agents, efforts must be made to investigate the mechanism(s) and kinetics of destruction of micro-organisms, enzymes, and nutrients by combinations of these agents. Such knowledge, coupled with multidisciplinary integration of advances in science, engineering, biology, chemistry, and nutrition can accelerate the introduction of various clean label, micro-biologically safe, and nutritious ESL foods.
This article is based on the Institute of Food Technologists Nonthermal Processing Division webcast titled “Extended Shelf-Life Foods—Ensuring Food Safety Through High Pressure–Based Technological Solutions” held Feb. 25, 2016. The authors thank Dr. Greg West, The Ohio State University for the helpful discussion. References to commercial products or trade names are made with the understanding that no endorsement or discrimination by The Ohio State University and Illinois Institute of Technology is implied.
A Technology That’s Right for the Times
Nonthermal high pressure processing (HPP)—which destroys pathogens without affecting taste or nutrient content—is proving to be a good fit for entrepreneurial companies working to deliver fresh, healthful products targeted to consumers who place a premium on clean label benefits.
August Vega, founder and CEO of Houston-based MALK Organics, maker of clean label vegan nut milks, opted for HPP because it allowed her to produce fresh refrigerated and nutrient-rich almond and pecan milks without using preservatives. Vega considered other pasteurization options but found that they didn’t deliver the fresh taste that HPP provided.
MALK (the name was chosen to convey the concept of “milk alternative”) made its retail debut late in 2014 and is currently sold in Kroger supermarkets nationwide and in Whole Foods Markets in the Southwest, Northeast, and Rocky Mountain regions. It is available in 12-ounce and 28-ounce bottles that retail for $4.99 and $6.99, respectively.
 For Austin Allen, the decision to use HPP on his four-item line of refrigerated gazpacho was driven primarily by the fact that, unlike thermal pasteurization processes, HPP has minimal effect on product flavor and texture. “For us, it really was about having a great-tasting product, so that’s why HPP was the only choice,” says Allen, founder and CEO of Tio Foods, New York, N.Y. As he prepared to launch his line of organic gazpacho, which made its debut in 2015, Allen explored other options, but quickly decided that HPP was the way to go. “As I was starting the company, I learned about HPP. I tried other products—[heat] pasteurized gazpachos, and they just didn’t taste the same.”
For Austin Allen, the decision to use HPP on his four-item line of refrigerated gazpacho was driven primarily by the fact that, unlike thermal pasteurization processes, HPP has minimal effect on product flavor and texture. “For us, it really was about having a great-tasting product, so that’s why HPP was the only choice,” says Allen, founder and CEO of Tio Foods, New York, N.Y. As he prepared to launch his line of organic gazpacho, which made its debut in 2015, Allen explored other options, but quickly decided that HPP was the way to go. “As I was starting the company, I learned about HPP. I tried other products—[heat] pasteurized gazpachos, and they just didn’t taste the same.”
A 4-ounce bottle of Tio gazpacho retails for $6.99. Distribution is currently concentrated in natural and specialty grocery retailers in the Northeast.
 Could HPP be a market disruptor in the baby food category, prompting a transition from shelf-stable jarred products to better-tasting refrigerated offerings? A recent blog on the website of HPP equipment supplier Hiperbaric makes the case for it, pointing to HPP’s ability to preserve heat-sensitive micronutrients, ensure food safety, and maintain favorable organoleptic properties.
Could HPP be a market disruptor in the baby food category, prompting a transition from shelf-stable jarred products to better-tasting refrigerated offerings? A recent blog on the website of HPP equipment supplier Hiperbaric makes the case for it, pointing to HPP’s ability to preserve heat-sensitive micronutrients, ensure food safety, and maintain favorable organoleptic properties.
Makers of refrigerated organic baby foods that employ HPP include Pure Spoon, Once Upon a Farm, and Little Spoon, among others. Mass merchandiser Target is currently testing the use of refrigerated endcap display fixtures in the baby food section, and Alyson Eberle founder of Austin, Texas–based Pure Spoon considers that a major step forward for baby food retailing. “We have to educate consumers, but we also have to educate stores” about the availability of refrigerated products, she notes.
 Some HPP users are doing their part to spread the word about the technology. Hope Hummus, Louisville, Colo., states on its website, for example, that “we only use cold pressure with state-of-the-art HPP technology to retain fresh, peak flavor, wonderful texture, and important nutrition without artificial preservatives.” And on its website, Los Angeles–based Nomva, which produces organic fruit and veggie purees, states, “we keep our packs safe and preserve natural nutrients by using high pressure processing.” As MALK’s Vega points out, “It’s about transparency and consumer education.”
Some HPP users are doing their part to spread the word about the technology. Hope Hummus, Louisville, Colo., states on its website, for example, that “we only use cold pressure with state-of-the-art HPP technology to retain fresh, peak flavor, wonderful texture, and important nutrition without artificial preservatives.” And on its website, Los Angeles–based Nomva, which produces organic fruit and veggie purees, states, “we keep our packs safe and preserve natural nutrients by using high pressure processing.” As MALK’s Vega points out, “It’s about transparency and consumer education.”
HPP is a process that matches up well with today’s consumer expectations, says Lisa Pitzer, marketing director for Avure Technologies, Erlanger, Ky., a supplier of HPP systems. “All HPP products are in the refrigerated space, so they’re in that segment—the outer perimeter [of the supermarket]—where consumers like to shop,” Pitzer notes.
—Mary Ellen Kuhn
V.M. Balasubramaniam, PhD, a professional member of IFT, is professor of food engineering, The Ohio State University, Columbus, Ohio (balasubramaniam.1@osu.edu). Ahmed E. Yousef, PhD, a professional member of IFT, is a professor of food microbiology, The Ohio State University, Columbus, Ohio (yousef.1@osu.edu). Jason Wan, PhD, a professional member of IFT, is associate director at the Institute for Food Safety and Health (IFSH) and professor and associate chair of food science and nutrition, Illinois Institute of Technology, Bedford Park, Ill. (jwan1@iit.edu). Ash Husain, PhD, a professional member of IFT, is chief technical officer, CedarLane Natural Foods, Carson, Calif. (ahusain@cedarlanefoods.com).
References
Anonymous. 2016. “Natural & Organic Retail Foods and Beverage Market Hits $69 Billion.” http://www.packagedfacts.com/about/release.asp?id=4006.
Balasubramaniam, V. M., D. Farkas, and E. J. Turek. 2008. “Preserving Foods through High Pressure Processing.” Food Technol. 62 (11): 32–38.
Brody, A. L. 2003. “Modified Atmosphere Packaging.” In Encyclopedia of Agricultural, Food, and Biological Engineering, edited by D. Heldman. 2nd ed., Taylor & Francis. doi: 10:1081/E-EAFE 1200007139.
Daryaei, H., V. M. Balasubramaniam, and J. D. Legan. 2013. “Kinetics of Bacillus cereus Spore Inactivation in Cooked Rice by Combined Pressure-Heat Treatment.” J. Food Prot. 76(4): 616–623. doi: 10.4315/0362-028X.JFP-12-447.
Eustice, R. F. and C. M. Bruhn. 2013. “Consumer Acceptance and Marketing of Irradiated Foods.” In Food Irradiation Research and Technology, edited by X. Fan and C. H. Sommers, 2nd ed., Blackwell Publishing.
Farkas, D. F., and D. G. Hoover. 2000. “High Pressure Processing.” J. Food Sci. 65(supplement s8): 47–64.
Federal Register. 2015. “Final Rule — Current Good Manufacturing Practice, Hazard Analysis, and Risk-Based Preventive Controls for Human Food.” https://www.federalregister.gov/documents/2015/09/17/2015-21920/current-good-manufacturing-practice-hazard-analysis-and-risk-based-preventive-controls-for-human.
FSA. 2008. “Food Standards Agency Guidance on Safety and Shelf-life of Vacuum and Modified Atmosphere Packed Chilled Foods with respect to non-proteolytic Clostridium botulinum.” Food Standards Agency. https://www.food.gov.uk/sites/default/files/multimedia/pdfs/publication/vacpacguide.pdf.
FDA. 2011. “Fish and Fishery Products Hazards and Controls Guidance,” 4th ed. http://www.fda.gov/downloads/Food/GuidanceRegulation/UCM251970.pdf.
IFSH. 2015. “IFSH Receives FDA Acceptance of Pressure Enhanced Sterilization Process for Commercial Production of Multi-Component Shelf-Stable Foods.” Food Safety, July 16. http://www.foodsafetymagazine.com/news/ifsh-receives-fda-acceptance-of-pressure-enhanced-sterilization-process-for-commercial-production-of-multicomponent-shelf-stable-foods/.
Legan, J. D., B. L. Chapman, and M. K. Bull. 2008. “Process for Reducing Spore Levels in Compositions.” World Patent WO 2008/083216.
Markland, S. M., D. F. Farkas, K. E. Kniel, and D. G. Hoover. 2013. “Pathogenic Psychrotolerant Sporeformers: An Emerging Challenge for Low-Temperature Storage of Minimally Processed Foods.” Foodborne Pathog. Dis. 10(5): 413–419. doi: 10.1089/fpd.2012.1355.
Martínez-Monteagudo, S. I., S. Kamat, N. Patel, G. Konuklar, N. Rangavajla, and V. M. Balasubramaniam. 2017. “Improvements in Emulsion Stability of Dairy Beverages Treated by High Pressure Homogenization: A Pilot-scale Feasibility Study.” J. Food Eng. 193: 42–52. doi: 10.1016/j.jfoodeng.2016.08.011.
Marth, E. H. 1998. “Extended Shelf Life Refrigerated Foods: Microbiological Quality and Safety.” Food Technol. 52(2): 57–62.
NACMCF. 2006. “Requisite Scientific Parameters for Establishing the Equivalence of Alternative Methods of Pasteurization.” J. Food Prot. 69(5): 1190–1216.
Nunes, K. 2016. “Clean label-a $180 billion global opportunity,” Food Business News, Oct. 6. http://www.foodbusinessnews.net/articles/news_home/Business_News/2016/10/Clean_label__a_$180_billion_gl.aspx?ID=%7B35B6F389-F481-4BF5-8DD1-9BAB90D5EA8B%7D&cck=1.
Peck, M. W., and S. C. Stringer. 2005. “The Safety of Pasteurized In-pack Chilled Meat Products with respect to the foodborne botulism Hazard.” Meat Sci. 70(3):461-475. doi: 10.1016/j.meatsci.2004.07.019.
Rodrigues, F. M., A. Rosenthal, J. H. Tiburski, and A. G. Cruz. 2015. “Alternatives to Reduce Sodium in Processed Foods and the Potential of High Pressure Technology.” Food Sci. Technol. (Campinas). 36(1): 1–8. doi: 10.1590/1678-457X.6833.
Silva, F. V. M. and P. A. Gibbs. 2010. “Non-proteolytic Clostridium botulinum Spores in Low-Acid Cold Distributed Foods and Design of Pasteurization Processes.” Trends Food Sci. Technol. 21(2): 95–105. doi: 10.1016/j.tifs.2009.10.011.
Skinner, G. E., K. M. Marshall, T. R. Morrissey, V. Loeza, E. Patazca, N. R. Reddy, and J. W. Larkin. 2014. “Combined High Pressure and Thermal Processing on Inactivation of Type E and Nonproteolytic Type B and F Spores of Clostridium botulinum.” J. Food Prot. 77(12): 2054–2061. doi: 10.4315/0362-028X.JFP-14-259.
Stewart , C. M., C. P. Dunne, and L. Keener. 2016. “Pressure-Assisted Thermal Sterilization Validation.” Chpt. 29. in High Pressure Processing of Food-Principles, Technology and Application, edited by V.M. Balasubramaniam, G. V. Barbosa-Canovas, and H. Lelieveld. New York: Springer LLC.
West, Greg. 2016. Extended Shelf Life Foods. Personal communication. The Ohio State Univ., Columbus, Ohio.
Zhang, H. Q., G. Barbosa-Canovas, V. M. Balasubramaniam, P. Dunne, D. Farkas, and J. Yuan, eds. 2011. Nonthermal Processing Technologies for Food. Chicago: IFT Press, Wiley-Blackwell Publishing.
Authors
-
V. M. Balasubramaniam, Ahmed E. Yousef, Jason Wan, , Ash Husain
Categories
-
Food Processing and Packaging
-
Nonthermal Processing
-
Food Technology Magazine



