FOOD SAFETY AND QUALITY
Monitoring Bacillus cereus in Dairy Products


Dairy product quality and safety greatly depend on the initial quality of the milk. © Image Source/DigitalVision/Getty Images
The quality and safety of dairy products greatly depends on the initial quality of the milk, since the prevalence of pathogens is likely to increase during milk processing. As a complex source of nutrients with high water activity and a near-neutral pH, milk provides an ideal growth environment for many microorganisms. The microbiota of raw milk are quite complex, and the types and concentrations of microorganisms vary based on animal health, hygiene and sanitation on farms, bedding, pasture lands, seasons, feedstuff, and a variety of other factors (Lund et al. 2000).
Even though milk is generally sterile while it is in the udder of a healthy cow, it can later become contaminated from the external surface of the udder or from other environments during milking, transportation, processing, and storage. The dairy industry ensures the safety and quality of milk by cooling raw milk to refrigeration temperatures until the milk is processed through pasteurization, ultra-high temperature treatment, and drying (Lund et al. 2000).
Sporeforming Bacteria Concerns
Among the microorganisms that enter milk through processing, sporeforming bacteria are a particular concern due to their ability to withstand adverse environmental conditions. Sporeformers, which are commonly found in the gastrointestinal tracts of animals and humans as well as in soil and aquatic environments, can be classified as thermophilic, mesophilic, or psychrotrophic. Thermophilic (heat-loving) sporeformers are more widespread in final products, and psychrotrophic (cold-tolerant) sporeformers grow while milk and milk products are refrigerated.
Sporeforming bacteria are gram-positive organisms encompassing nine genera: Alicyclobacillus, Amphibacillus, Bacillus, Clostridium, Desulfotomaculum, Filobacillus, Geobacillus, Sporolactobacillus, and Sulfolobus. Hundreds of species under the genera can form endospores that make the species resistant to conditions such as high pressures, extreme heat, freezing, desiccation, starvation, chemicals, and radiation. Anaerobic sporeforming bacteria of the Clostridium genus, especially C. botulinum and C. perfringens, are of public health significance. C. botulinum produces an array of neurotoxins, the most potent toxins known to humans. C. perfringens produces a diarrheal enterotoxin as well as other extracellular toxins (Matthews et al. 2017).
The aerobic sporeforming bacteria of the Bacillus genus, B. cereus in particular, impact both food quality and food safety. The Bacillus genus, which is likely the oldest and most diverse genus of bacteria, has diverse ecology, and its species inhabit many natural environments, including soil and water (Matthews et al. 2017).
B. cereus and Foodborne Illness
B. cereus is a gram-positive, rod-shaped, motile, sporeforming, opportunistic pathogen usually found in soil, where vegetative cells sporulate to form spores under adverse conditions, and the spores germinate to become vegetative cells under favorable conditions. Select B. cereus strains may live in the gastrointestinal tracts of invertebrates and small rodents. The spores, usually present at 105/g soil, can readily spread to cows’ udders, raw milk, roots, tubers, and tops of crops and then enter human food or animal feed supplies. Heat-resistant spores may become enriched in foods that are thermally processed (Matthews et al. 2017).
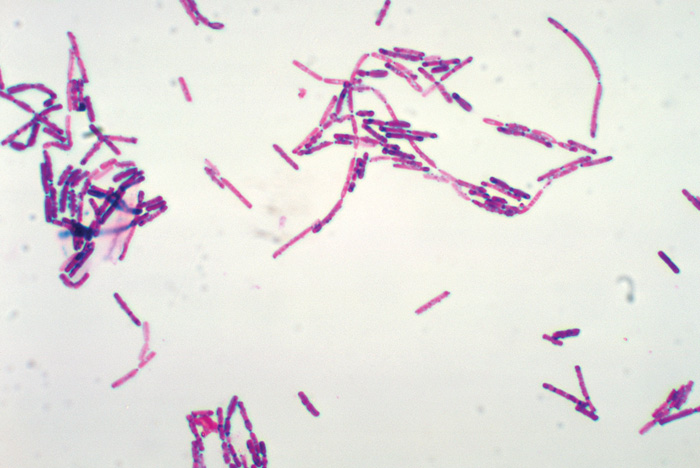
Chains of gram-positive Bacillus cereus cells are shown under a light microscope. Photo courtesy of Centers for Disease Control and Prevention/Dr. William A. Clark
Some species of the B. cereus group, also known as B. cereus sensu lato (s.l.), cause foodborne illness in humans. Until recently, the group has comprised nine closely related species (Liu et al. 2015): B. anthracis, B. cereus sensu sitricto (s.s.), B. thuringiensis, B. mycoides, B. pseudomycoides, B. weihenstephanensis, B. cytotoxicus, B. toyonensis, and B. widemanni. Since 2017, the B. cereus group has been expanded with nine new species isolated from marine sediments: B. paranthracis, B. pacificus, B. tropicus, B. albus, B. mobilis, B. luti, B. proteolyticus, B. nitratireducens, and B. paramycoides (Liu et al. 2017). The B. cereus group designation is practical because differ-
entiating among several species of the B. cereus group can be difficult.
B. cereus sensu sitricto (s.s.)—or B. cereus, as it is commonly called—has been linked to two types of foodborne illness: emetic and diarrheal. Emetic illness is caused by the emetic toxin cereulide, produced by vegetative cells growing in the contaminated food. It causes nausea and vomiting within 0.5–6 hr of ingestion. The total number of B. cereus cells needed to cause emetic illness is estimated to be 105–108/g food (Matthews et al. 2017).
Diarrheal illness is caused by at least three heat-labile enterotoxins: hemolysin BL, nonhemolytic enterotoxin, and cytotoxin K. Vegetative cells, ingested as viable cells or spores, produce enterotoxin(s) in the small intestine, causing diarrhea, cramps, and rectal tenesmus in 6–14 hr. The infective dose of the diarrheal illness is estimated to range between 105 and 108 cells (Matthews et al. 2017).
A few B. cereus strains have been reported to produce both the emetic toxin and enterotoxin(s), causing both emetic and diarrheal illnesses. B. cereus foodborne illness cases in the United States are estimated to be ~63,400 per yr, but these illnesses likely are greatly underestimated due to their mild, brief (usually <24 hr), and self-limiting nature; the symptoms shared with other foodborne illnesses; and the possibility that illness caused by>B. cereus-contaminated milk can be limited to 1–2 cases, which would not be considered an outbreak (Matthews et al. 2017).
Pinpointing B. cereus Contamination
Because of the B. cereus group’s omnipresence in nature, it is highly challenging to determine definite sources of contamination by the group on dairy farms. The most common contamination sources include soil, bedding, feces, feed, silage, water, and milking equipment.

Soil and feed are two of the most common B. cereus contamination sources. © ahavelaar/iStock/Getty Images Plus
In addition, the composition of Bacillus microbiota in raw milk exhibits seasonal differences. Higher B. cereus counts are observed in summer months due in part to pasturing, while feed and bedding are the leading sources of B. cereus contamination in winter months (Lund et al. 2000). In a study conducted in Belgium, members of the B. cereus group were isolated more often in late summer/early fall and more often in milk from organic dairy farms when compared with milk from conventional dairy farms (Coorevits et al. 2008).
When B. cereus contaminates raw milk, the entire milk processing continuum is affected because B. cereus strains can survive the pasteurization process due to production of heat-resistant spores; the strains involved in foodborne illness have higher heat resistances (D95oC of ~24 min) (Matthews et al. 2017). The prevalence rate of B. cereus in raw milk is somewhat high, although it is typically at low levels (<1 and up to 10–100 spores ml raw milk), but processed milk products such as pasteurized milk, cheeses, or milk powder can include 10>2–103 spores/g (Logan and De Vos 2011).
B. cereus spores also are highly hydrophobic and contain appendages and/or pili, which allows them to aggregate and adhere to inert surfaces (e.g., stainless steel) to form biofilms in dairy processing environments. These biofilms —present on or in dead ends, corners, cracks, crevices, gaskets, valves, joints, etc.—can resist cleaning and sanitation and then detach and disperse, leading to product contamination. To combat this, the dairy industry needs to maintain rigorous biofilm monitoring and control strategies: cleaning-in-place processes, biocides, and other methods, including ozone, ultrasonication, and bacteriophage (Unlu 2020, Gopal et al. 2015).
When B. cereus contaminates raw milk, the entire milk processing continuum is affected because B. cereus strains can survive the pasteurization process due to production of heat-resistant spores.
B. cereus contamination of milk powder is also a major food safety concern. Milk powders can be stored for extended periods of time due in part to their low water activity, but bacterial spores can remain dormant until more favorable conditions are present, and then spore germination and outgrowth can ensue (Driks and Eichenberger 2016, Setlow 2014).
Powdered infant milk formula (IMF), which is produced to closely mimic the composition of human milk, influences the development of the gut microbiota in infants, who have underdeveloped immune systems. This means IMF needs to be screened for select foodborne pathogenic bacteria, including B. cereus (Forsythe 2005). Di Pinto et al. (2013) identified 11 IMF samples out of 60 samples as positive for Bacillus species, and five of these samples were characterized as B. cereus. Lequin et al. (2005) studied fatal hemorrhagic meningoencephalitis caused by B. cereus infections in three preterm infants. While the source(s) of these infections were unknown, it is well-known that IMF can harbor B. cereus.
Impact on Quality
In addition to its impact on microbial safety, the genus Bacillus can impact the quality of milk and milk products. Due to its enzymatic activities, B. cereus can cause off-flavors such as bitty cream and sweet curdling in milk and milk products. Bitty cream—the floating of clumps of fat—is caused by lecithinase activity, while the sweet curdling defect—the curdling of milk in the absence of any acidifying agent—is caused by proteolytic enzyme activity (Gopal et al. 2015).
The overall prevalence of B. cereus in dairy products is difficult to estimate. Although preventive measures have been designed to control microbial contamination of dairy products in the past century (Boor et al. 2017), B. cereus species contamination continues to be reported in various products all over the world (Liu et al. 2020, Pedonese et al. 2019, Rossi et al. 2018, Yusuf et al. 2018, Zhou et al. 2008, Ombui et al. 2005). With global increases in demand for and consumption of dairy foods and dairy-based ingredients, it is fundamental that the dairy industry continue to meet quality and safety standards, with particular attention to B. cereus spores in dairy foods. Fittingly, novel analytical methods for spore detection—LAMP, electrochemical sensors, lateral flow tests—have been getting attention in the dairy industry due to their simplicity and sensitivity, although there are limitations in their commercial availability (Vidic et al. 2020).
REFERENCES
Boor, K. J., M. Wiedmann, S. Murphy, et al. 2017. “A 100-Year Review: Microbiology and Safety of Milk Handling.” J. Dairy Sci. 100(12): 9933–9951. doi: 10.3168/jds.2017-12969.
Coorevits, A., V. De Jonghe, J. Vandroemme, et al. 2008. “Comparative Analysis of the Diversity of Aerobic Spore-Forming Bacteria in Raw Milk from Organic and Conventional Dairy Farms.” Sys. Appl. Microbiol. 31(2): 126–140. doi: 10.1016/j.syapm.2008.03.002.
Di Pinto, A., E. Bonerba, G. Bozzo, et al. 2013. “Occurrence of Potentially Enterotoxigenic Bacillus cereus in Infant Milk Powder.” Eur. Food Res. Technol. 237: 275–279. doi: 0.1007/s00217-013-1988-8.
Driks, A. and P. Eichenberger, eds. 2016. The Bacterial Spore: From Molecules to Systems. Washington, D.C.: ASM Press.
Forsythe, S. J. 2005. “Enterobacter sakazakii and Other Bacteria in Powdered Infant Milk Formula.” Mat. Child Nut. 1: 44–50. doi: 10.1111/j.1740-8709.2004.00008.x.
Gopal, N., C. Hill, P. R. Ross, et al. 2015. “The Prevalence and Control of Bacillus and Related Spore-Forming Bacteria in the Dairy Industry.” Front. Microbiol. 6: 1418. doi: 10.3389/fmicb.2015.01418.
Lequin, M. H., J. R. Vermeulen, R. M. van Elburg, et al. 2005. “Bacillus cereus Meningoencephalitis in Preterm Infants: Neuroimaging Characteristics.” Am. J. Neuroradiol. 26(8): 2137–2143.
Liu, Y., Q. Lai, M. Göker, et al. 2015. “Genomic Insights into the Taxonomic Status of the Bacillus cereus Group.” Sci. Rep. 5: 14082. doi: 10.1038/srep14082.
Liu, Y., J. Du, Q. Lai, et al. 2017. “Proposal of Nine Novel Species of the Bacillus cereus Group.” Int. J. Syst. Evol. Microbiol. 67: 2499–2508. doi: 10.1099/ijsem.0.001821.
Liu, X.-Y., Q. Hu, F. Xu, et al. 2020. “Characterization of Bacillus cereus in Dairy Products in China.” Toxins 12(7): 454. doi: 10.3390/toxins12070454.
Logan, Niall A. and P. De Vos, eds. 2011. Endospore-Forming Soil Bacteria: Soil Biology. Berlin-Heidelberg: Springer-Verlag.
Lund, B. M., T. C. Baird-Parker, and G. W. Gould, eds. 2000. Microbiological Safety and Quality of Food. New York: Springer.
Matthews, K. R., K. E. Kniel, and T. J. Montville. 2017. Food Microbiology: An Introduction. Washington, D.C.: ASM Press.
Ombui, J. N. and J. G. Nduhiu. 2005. “Prevalence of Enterotoxigenic Bacillus cereus and its Enterotoxins in Milk and Milk Products in and Around Nairobi.” East Afr. Med. J. 82: 280–284. doi: 10.4314/eamj.v82i6.9297.
Pedonese, F., G. Verani, B. Torracca, et al. 2019. “Effect of an Italian Propolis on the Growth of Listeria monocytogenes, Staphylococcus aureus and Bacillus cereus in Milk and Whey Cheese.” Ital. J. Food Saf. 8: 8036. doi: 10.4081/ijfs.2019.8036.
Rossi, G. A. M., H. O. Silva, C. E. G. Aguilar, et al. 2018. “Comparative Genomic Survey of Bacillus cereus sensu stricto Isolates From the Dairy Production Chain in Brazil.” FEMS Microbiol. Lett. 365(3). doi: 10.1093/femsle/fnx283.
Setlow, P. 2014. “Germination of Spores of Bacillus Species: What We Know and Do Not Know.” J. Bacteriol. 196: 1297–1305. doi: 10.1128/JB.01455-13.
Ünlü, G. 2020. “Bacterial Biofilms: Formation, Prevention, and Control.” Food Technol. 74(10): 73–76.
Vidic, J., C. Chaix, M. Manzano, et al. 2020. “Food Sensing: Detection of Bacillus cereus Spores in Dairy Products.” Biosensors 10(3): 15. doi: 10.3390/bios10030015.
Yusuf, U., S. K. Kotwal, S. Gupta, et al. 2018. “Identification and Antibiogram Pattern of Bacillus cereus From the Milk and Milk Products in and Around Jammu Region.” Vet. World 11: 186–191. doi: 10.14202/vetworld.2018.186-191.
Zhou, G., H. Liu, J. He, et al. 2008. “The Occurrence of Bacillus cereus, B. thuringiensis and B. mycoides in Chinese Pasteurized Full Fat Milk.” Int. J. Food Microbiol. 121(2): 195–200. doi: 10.1016/j.ijfoodmicro.2007.11.028.
Authors
-
Gülhan Ünlü
Categories
-
Food Categories
-
Food Safety and Defense
-
Pathogens
-
Foodborne Illness
-
Dairy
-
Food Quality
-
Quality Assurance and Quality Control
-
Food Technology Magazine



