As the food marketplace becomes more global, it becomes increasingly important to understand how low-calorie sweeteners are regulated in the U.S. and internationally.
Regulatory Status of Alternative Sweeteners
Alternative sweeteners are among the most thoroughly studied and scrupulously regulated ingredients in our food supply. They are also the most visible. Consumers are very familiar with sweeteners—they understand what sweeteners do and know them individually by their trade names, if not generically.

In a 2004 nationally projectable consumer survey conducted by the Calorie Control Council, 84% of respondents reported using low-calorie, reduced-sugar, or sugar-free foods and beverages. And 85% of these users said they were interested in being offered additional light products.
Food product developers today have a number of sweeteners from which to choose. Having a variety of sweeteners available is important because no sweetener alone, including sucrose, is perfect for all uses. But with several available, each sweetener can be used in the applications for which it is best suited, and manufacturers can overcome limitations of individual sweeteners by using them in blends. Also, most sweeteners when combined have a synergistic effect, in which the sweetness of the combination is greater than the sum of the individual parts.
Five low-calorie sweeteners and eight polyols (sugar alcohols) are currently approved for use in the United States. Additional low-calorie sweeteners are approved in other parts of the world. With the ever-increasing global marketplace it is important to understand how sweeteners are regulated both nationally and internationally.
Regulatory Terms
For understanding the regulatory status of sweeteners, a few terms are especially important:
• Acceptable Daily Intake (ADI). This is an important concept, both nationally and internationally, but one that is frequently misunderstood. The Food and Drug Administration defines the ADI as the amount of a compound that can be safely consumed each day on a chronic basis. The agency has further stated that the ADI is a conservative estimated safe exposure level, which anticipates a continuous lifetime exposure. It is used by FDA as an internal benchmark to evaluate the actual intake of a substance, and as an aid in reviewing possible additional uses for a food ingredient. The ADI is expressed in milligrams per kilogram of body weight.
The ADI is determined by toxicological testing in animals, and sometimes humans, and is normally estimated by applying an intentionally conservative safety factor, generally 100-fold. For example, if studies of a particular substance at a dose level of 1,000 mg/kg showed no effect, an ADI for that substance using the 100-fold safety factor would be 10 mg/kg. The ADI does not represent a maximum allowable daily intake level, and it should not be regarded as a specific point at which safety ends and possible health concerns begin. In fact, FDA has said that it is not concerned if consumption occasionally exceeds the ADI.
Using aspartame as an example, however, to reach the 50 mg/kg ADI set by FDA for aspartame, a 150-lb person would have to consume 20 12-oz cans of soft drink every day for a lifetime. Extensive market research has shown that aspartame consumption patterns for the general population and various subgroups are well below the ADI. Aspartame consumption by high-level consumers (90th percentile) in the general population, including children, is 5–10% of the ADI. This means that 9 out of 10 people consume less than 10% of the ADI.
--- PAGE BREAK ---
• General-Purpose Approval. This FDA term means that the sweetener may be used in any product where a standard of identity does not preclude such use. For example, a standard might call for the use of a nutritive carbohydrate sweetener. Therefore, acesulfame potassium could not be used because it is neither nutritive nor a carbohydrate.
• GRAS. According to federal law and regulations, any substance that is “Generally Recognized as Safe” for a particular use or uses may be used in foods for that purpose without premarket approval from FDA. Although a GRAS determination is a more expedient way to bring a food ingredient to market than the food additive petition process, general recognition of safety calls for the same quantity and quality of scientific evidence as would be required to obtain a food additive regulation for a substance.
Scientific procedures appropriate to establish the safety of a substance include human, animal, analytical, and other studies. General recognition of safety through scientific procedures ordinarily must be based on published studies, although those studies can be bolstered by unpublished evidence. The term “safe” means that there is reasonable certainty in the minds of competent scientists that a substance is not harmful under intended conditions of use.
FDA is now using a “GRAS notification system,” under which a manufacturer may notify the agency that it has determined through self-affirmation that its substance is GRAS, providing evidence supporting its decision. After evaluating the notification and agreeing that the evidence submitted supports a GRAS determination, FDA issues a letter notifying the manufacturer that the agency has no questions at the time concerning the company’s conclusion that the substance is GRAS under the intended conditions of use. Or FDA may request additional information or inform the manufacturer that the notice does not provide a sufficient basis for a determination that the substance is GRAS under the conditions of its intended use. GRAS notifications and FDA responses are available on FDA’s Web site at www.cfsan.fda.gov/ ~rdb/opa-gras.html. A notification to which FDA originally responded that the agency has no concerns may be revisited if new information indicates a reason for concern.
The cost in both time and money for the approval of a new food additive is almost prohibitive, resulting in an increased number of GRAS notifications and a decrease in the number of food additive petitions.
Regulatory Status
The regulatory status of the low-calorie (intense) sweeteners and polyols varies by country.
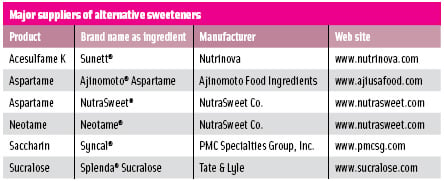 • Intense Sweeteners. The five low-calorie, intense sweeteners approved for use in the U.S. are acesulfame potassium, aspartame, neotame, saccharin, and sucralose. Acesulfame potassium, aspartame, neotame, and sucralose have general-purpose approval. Food additive petitions for alitame and cyclamate are pending before FDA. Cyclamate, though not approved in the U.S., is approved in more than 50 countries and may be imported into the U.S. to make products for export. Saccharin is approved in the U.S. for special dietary purposes, such as to provide sweetness in reduced-calorie products.
• Intense Sweeteners. The five low-calorie, intense sweeteners approved for use in the U.S. are acesulfame potassium, aspartame, neotame, saccharin, and sucralose. Acesulfame potassium, aspartame, neotame, and sucralose have general-purpose approval. Food additive petitions for alitame and cyclamate are pending before FDA. Cyclamate, though not approved in the U.S., is approved in more than 50 countries and may be imported into the U.S. to make products for export. Saccharin is approved in the U.S. for special dietary purposes, such as to provide sweetness in reduced-calorie products.
With the exception of neotame, all of the above sweeteners are approved in the European Union. Acesulfame potassium, aspartame, and sucralose are approved in Canada. Health Canada has issued a proposal to approve neotame and a stakeholders consultation document noting that it plans to approve saccharin for use in foods and beverages. Saccharin is currently available in tabletop sweetener, cosmetics, and pharmaceuticals in Canada. These sweeteners, and others to be mentioned later, are approved in numerous other countries as well.
• Polyols. The eight polyols approved for use in the U.S. are erythritol, isomalt, lactitol, maltitol, mannitol, polyglycitols (hydrogenated starch hydrolysates), sorbitol, and xylitol. Xylitol is approved for use in special dietary foods; mannitol is approved for use in specific products at specific levels; and sorbitol has been affirmed as GRAS by FDA. Erythritol, isomalt, lactitol, maltitol, and polyglycitols are self-affirmed as GRAS with no questions pending from FDA.
--- PAGE BREAK ---
Polyols are important sweeteners for the production of reduced-calorie and sugar-free foods and beverages. They provide the bulk of sugar with fewer calories than the 4 kcal/g contributed by sugar. FDA allows the use of the following caloric values for the polyols: 0.2 kcal/g for erythritol, 1.6 for mannitol, 2.0 for isomalt and lactitol, 2.1 for maltitol, 2.4 for xylitol, 2.6 for sorbitol, and 3.0 for polyglycitols or hydrogenated starch hydrolysates.
Products sweetened with polyols or a combination of polyols and low-calorie sweeteners may, under FDA regulations, carry a “Does not promote tooth decay” label.
Approval of Sweeteners
Food ingredients are evaluated and/or regulated by numerous national and international bodies in addition to FDA. International groups include the Food and Agriculture Organization/World Health Organization’s Joint FAO/ WHO Expert Committee on Food Additives (JECFA) and the Codex Alimentarius Commission.
• JECFA is an international group of experts who serve, without remuneration, in their personal capacities rather than as representatives of their governments or other bodies. Members are selected primarily for their ability and technical experience, with consideration given to adequate geographical distribution. JECFA’s reports contain the collective views of the group and do not necessarily represent the decision or the stated policy of WHO or FAO. The experts convene to give advice on technical and scientific matters, establish specifications for identity and purity of food additives, evaluate the toxicological data, and recommend, where appropriate, ADIs for humans. JECFA also acts in an advisory capacity for the Codex Committee on Food Additives.
All the sweeteners discussed above have been reviewed, deemed safe, and assigned an ADI by JECFA. For example, JECFA has assigned an ADI “not specified,” the most favorable assignation, to each of the polyols mentioned; this means that, on the basis of the available data (chemical, biochemical, toxicological, and other), the total daily intake of the substance, arising from its use at the levels necessary to achieve the desired effect and from its acceptable background in food, does not, in the opinion of the committee, represent a hazard to health. For that reason and for reasons stated in the individual evaluations, the establishment of an ADI expressed in a numerical form is deemed not necessary.
Countries that do not have the review capability of the U.S., the EU, Canada, and Japan depend on JECFA for scientific evaluation of food additives. In addition to its evaluation of all sweeteners mentioned above, JECFA in 2004 allocated a temporary ADI to steviol glycosides, sweet compounds in stevia. To remove the “tentative” status, JECFA requested human studies on the pharmacological effects of steviol glycosides in humans from repeated exposure to dietary and therapeutic doses in normotensive and hypotensive individuals and in insulin-dependent and insulin-independent diabetics. The information was requested by 2007, and JECFA is scheduled to again review steviol glycosides at its June 2007 meeting. Although stevioside can currently be used in the U.S. as a food supplement, it is not approved as a sweetener. Should JECFA assign the steviol glycosides a permanent ADI, this would likely lead to efforts for approval in the U.S.
• The Codex Alimentarius Commission is intended to guide and promote the elaboration and establishment of definitions and requirements for foods, including food additives, and to assist in their harmonization and thereby facilitate trade. The Codex Committee for Food Additives and Contaminants (CCFA) is charged with establishing or endorsing permitted maximum or guideline levels for individual food additives. CCFA is developing a General Standard for Food Additives (GSFA) that lists food additives that have been reviewed and assigned an ADI (either numerical or “not specified”) by JECFA. The GSFA provides a list of food categories for which an additive may be used and the levels of use for each category. Acesulfame potassium, alitame, aspartame, cyclamate, neotame, saccharin, sucralose, and the aspartame–acesulfame salt were scheduled to be addressed at CCFA’s late April 2007 meeting.
--- PAGE BREAK ---
CCFA has before it a proposal for a horizontal approach to the use of intense sweeteners. This approach, if approved, would list categories in which such sweeteners could be used; intense sweeteners could not be used in categories not appearing on this list (e.g., infant formula). With eight sweeteners and approximately 70 categories in which the sweeteners might be used, it appears unlikely that CCFA will complete its evaluation of sweeteners this year. An Electronic Working Group (eWG) on the GSFA has been working for more than six months on the food additives to be evaluated at the April meeting; they include not only the eight sweeteners but also more than 25 other additives. The eWG’s recommendations include requests for additional information, such as technological need, for a number of categories.
Other Sweetener Alternatives
Other alternatives to sucrose currently available for use in the U.S. and some other countries include the nutritive sweeteners isomaltulose, trehalose, and tagatose. FDA has issued letters stating that it has no questions at this time in response to GRAS notifications for each of them.
Isomaltulose, providing 4 kcal/g, has a sweetness similar to that of sugar, is less cariogenic than sucrose, and is low glycemic.
Trehalose also provides 4 kcal/g, but its sweetness may be less intense and more persistent than that of sucrose. Interestingly, trehalose was approved by the Ministry of Agriculture, Fisheries and Food in the United Kingdom in 1991 as a cryoprotectant for freeze-dried foods—it appears to stabilize proteins against damage caused by drying and freezing.
Tagatose is almost as sweet as sugar, noncariogenic and, like most sweeteners, synergistic with other sweeteners. FDA allows a claim of 1.5 kcal/g for tagatose, which provides the bulk of sugar.
Alternative sweeteners individually and as a group are the most thoroughly researched ingredients in the food supply. The safety of the approved sweeteners has been evaluated and confirmed by numerous regulatory and scientific bodies time and time again.
Low-calorie products continue to be in demand, and consequently so are the ingredients that make them possible. Taste remains king, and the approval of multiple alternative sweeteners facilitates the development of more and more good-tasting products. With obesity increasing in many parts of the world and consumers becoming increasingly aware that “calories still count,” the number of successful light products containing alternative sweeteners should soar.
Sources for More Information
The Calorie Control Council, a nonprofit international association of manufacturers of low-calorie, reduced-fat, and light foods and beverages, offers the following Web sites:
www.acesulfame.org
www.aspartame.org
www.saccharin.org
www.sucralose.org
www.polyol.org
www.caloriecontrol.org
Other information on regulatory matters can be found on the following Web sites:
Codex Alimentarius Commission:
www.codexalimentarius.net/web/index_en.jsp
Joint FAO/WHO Expert Committee on Food Additives: www.codexalimentarius.net/web/jecfa.jsp
Food and Drug Administration:
www.cfsan.fda.gov
How to Submit a GRAS Notice:
www.cfsan.fda.gov/~dms/opafrgr.html
Summary of All GRAS Notices:
www.cfsan.fda.gov/~rdb/opagras.html
Code of Federal Regulations,Title 21:
www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/cfrsearch.cfm
by Lyn O’Brien Nabors, a Professional Member of IFT, is President, Calorie Control Council, 1100 Johnson Ferry Rd., Suite 300, Atlanta, GA 30342 (lnabors@kellencompany.com).
Authors
-
LYN O'BRIEN NABORS
Categories
-
Food Business Trends
-
Food Health Nutrition
-
Food Ingredients and Additives
-
Food Policy
-
Sugars and Sweeteners
-
Global Markets and Trade
-
Food Technology Magazine



