GRAS Flavoring Substances 24
[DUE TO THE AMOUNT OF GRAPHICS IN THIS ARTICLE, IT IS ADVISED THAT YOU DOWNLOAD THE PDF VERSION FOR BETTER READABILITY]
It is now half of a century since passage of the 1958 Food Additives Amendment (FAA) to the Federal Food, Drug, and Cosmetic Act—Public Law 85-929, 72 Stat. 1784 (1958), codifi ed at 21 U.S.C. Sec. 348 (1988)—that exempted from food additive status those substances “generally recognized as safe” (GRAS) under conditions of intended use. In order for the use of a substance to qualify for GRAS status, the safety of the substance must be evaluated by experts qualified by training and experience and adequately shown through scientific procedures to be “generally recognized as safe.” For almost five decades, the Expert Panel of the Flavor and Extract Manufacturers Association (FEMA) has maintained an independent program to evaluate the safety of substances for their intended use as flavor ingredients. During that time, more than 2,600 flavor ingredients achieved GRAS status, approximately 200 candidates were withdrawn from GRAS consideration due to lack of available data or safety concerns, and 11 substances had their previous GRAS status revoked (or “DeGRASed”) due to specific safety concerns or an absence of requested safety data.
 At the beginning of the FEMA GRAS Program, FEMA flavor manufacturers submitted data on approximately 1,400 flavoring substances that had been in commerce in the United States at the time the 1958 Amendment was adopted. In the first round of safety evaluations, the Expert Panel systematically considered data on these chemically defined substances and concluded that 1,124 of these flavor ingredients were GRAS under conditions of their intended use in food flavorings. In accordance with the provisions of the 1958 Amendment that the use of these substances be “generally recognized as safe,” the Expert Panel published the first list of GRAS flavoring substances in 1965 (Hall and Oser, 1965). This began a tradition of periodic publication of GRAS lists of flavoring substances, including their principal name, synonyms, and their recommended average added usual and maximum use levels. In addition, there was a desire to publish the background scientific data upon which GRAS deliberations were based. Under contract to the U.S. Food and Drug Administration (FDA), relevant safety data were compiled on approximately 1,300 substances and published in a series of Scientific Literature Reviews (SLRs) between 1974 and 1979. Each SLR was organized into groups of structurally related flavoring ingredients that participated in common pathways of metabolism and exhibited similar toxicological potential.
At the beginning of the FEMA GRAS Program, FEMA flavor manufacturers submitted data on approximately 1,400 flavoring substances that had been in commerce in the United States at the time the 1958 Amendment was adopted. In the first round of safety evaluations, the Expert Panel systematically considered data on these chemically defined substances and concluded that 1,124 of these flavor ingredients were GRAS under conditions of their intended use in food flavorings. In accordance with the provisions of the 1958 Amendment that the use of these substances be “generally recognized as safe,” the Expert Panel published the first list of GRAS flavoring substances in 1965 (Hall and Oser, 1965). This began a tradition of periodic publication of GRAS lists of flavoring substances, including their principal name, synonyms, and their recommended average added usual and maximum use levels. In addition, there was a desire to publish the background scientific data upon which GRAS deliberations were based. Under contract to the U.S. Food and Drug Administration (FDA), relevant safety data were compiled on approximately 1,300 substances and published in a series of Scientific Literature Reviews (SLRs) between 1974 and 1979. Each SLR was organized into groups of structurally related flavoring ingredients that participated in common pathways of metabolism and exhibited similar toxicological potential.
These reviews served as the foundation for the fi rst comprehensive re-evaluation and affi rmation of the GRAS status of more than 1,740 flavor ingredients in a program known as GRAS affirmation (GRASa). The GRASa Program began in 1975 and was completed in 1985. In the next 10-year period (1985–1995), relatively few new substances (approximately 75) were GRASed for use as flavoring substances, suggesting that there may be an upper limit to the number of commercially viable flavoring substances.
However, two mutually connected initiatives drastically altered the rate of GRAS evaluations. First, in 1994, the Expert Panel began a second comprehensive evaluation of the safety of all chemically defined GRAS fl avoring substances in a program designated GRAS reaffirmation (GRASr). Second, in conjunction with FDA, the flavor industry supported a new, more-comprehensive initiative to expedite the global evaluation of all existing flavoring substances. After more than 30 years of evaluating the safety of individual flavoring substances, the World Health Organization and United Nations Food and Agriculture Organization Joint Expert Committee on Food Additives (JECFA) adopted a novel flavor evaluation procedure in 1995 that allowed for the systematic evaluation of chemical groups of fl avoring ingredients beginning in 1996. These chemical groups of fl avor ingredients had been previously evaluated by the FEMA Expert Panel in the GRASr program. Every year since 1996, FDA has requested that the Codex Committee on Food Additives (CCFA) place groups of GRAS fl avor ingredients on the JECFA agenda for evaluation at its annual meeting. As of 2008, JECFA has evaluated more than 1,750 substances and concluded that these flavoring agents are “safe under current conditions of intake.” The JECFA list is updated annually following each JECFA meeting.
The consistent conclusions rendered by the FEMA Expert Panel and JECFA have, to a considerable extent, created an open positive list of flavoring substances for use in the U.S. and in WHO member countries that wish to adopt the FEMA and/or JECFA lists. In other industrialized regions, such as the European Union (EU) and Japan, open positive lists of flavoring substances either are being developed or are currently in existence. In the EU, a positive list is being compiled based on the safety evaluations performed by JECFA prior to 2000 and thereafter by the European Food Safety Authority. In Japan, the Ministry of Health, Labor, and Welfare maintains a list of currently approved fl avoring substances, and recent safety evaluations of specific groups of flavor ingredients (e.g., pyrazine derivatives) have added new groups of structurally related substances to the existing list.
As the JECFA list has developed, flavor manufacturers in these other industrialized regions (Europe and Asia) have, quite understandably, taken an interest in having existing flavoring ingredients specific to their regions added to the FEMA and JECFA lists. Hence, both EU and Japanese flavor manufacturers have, through their respective international and national trade associations, submitted these substances for safety evaluation, first by the FEMA Expert Panel and then by JECFA. As a result, there has been renewed interest in the GRAS Program.
--- PAGE BREAK ---
Beginning with the publication of GRAS 18 in 1998, there has been a significant increase in the number of substances GRASed annually. GRAS 22 (published in 2005) and GRAS 23 (published in 2007) contain 185 and 175 new flavor ingredients, respectively. The vast majority of GRAS applications for substances in GRAS 22 and 23 were submitted on behalf of EU and Japanese manufacturers. In this publication (i.e., GRAS 24), 236 new flavor ingredients, mostly of Japanese origin, have been granted GRAS status. Therefore, in a four-year span, the GRAS list of chemically defined flavor ingredients has increased by approximately 25%. It is anticipated that the majority of flavor ingredients historically used in these regions have now been recognized as GRAS and future GRAS lists will contain mainly flavor ingredients resulting from global manufacturer research and development pipelines.
Genotoxicity Assays and Flavor Safety Evaluation
The Panel has now concluded its second comprehensive re-evaluation (GRASr) of the current safety data related to the use of GRAS substances as flavor ingredients. Because these evaluations were performed on groups of structurally related substances, it provided the Panel with the opportunity to screen robust datasets that included in vitro and in vivo genotoxicity assays. The Panel considered the relevance of this genotoxicity data in the context of existing information on the biochemical fate of the fl avor ingredients and animal toxicology and carcinogenicity. For example, for a group of structurally related benzyl derivatives (e.g., benzyl alcohol, benzaldehyde, benzoic acid, benzyl acetate), the Panel was able to compare the results of more than 100 in vitro and 17 in vivo genotoxicity assays with in vivo data on metabolism, short– and long-term toxicity, and lifetime carcinogenicity studies for five principal flavor ingredients in the group (Adams et al., 2005). Although there were isolated reports of clastogenicity or mutations in in vitro chromosomal aberration, sister chromatid exchange, and mouse lymphoma assays, these studies could not be confirmed in repeat assays. Additionally, there was no evidence of genotoxicity in any of the 17 in vivo assays performed on the same members of the group of benzyl derivatives. When these data are combined with the observations that benzyl derivatives 1) undergo high-capacity enzyme-catalyzed oxidative metabolism in animals to yield benzoic acid derivatives that are readily excretable as glycine conjugates, 2) show low oral toxicity in short– and long-term studies in mice and rats, and 3) exhibit no evidence of carcinogenicity relevant to human health, it can be concluded that the few isolated reports of a genotoxic response in vitro are not relevant to the safety of these substances when consumed at low levels as added flavoring substances.
During the past 15 years, the Panel has evaluated safety data, including genotoxicity data, on more than 50 groups of structurally related flavoring substances. Two chemical groups, the ,s-unsaturated aldehydes (Adams et al., 2008) and heterocyclic -ketoenols (JECFA, 2006) are mutagenic in the reverse bacterial assays in some strains of Salmonella typhimurium (Ames TA100, TA102, and TA104) and in some mammalian cell forward mutation assays (mouse lymphoma), and show evidence of clastogenicity in the sister chromatid exchange and chromosomal aberration assays. In a majority of in vitro genotoxicity assays involving ,s-unsaturated aldehydes, high concentrations (20 to 40 μM) in cell lines poor in detoxication capacity provide opportunity for either direct interaction of the ,s-unsaturated aldehyde with DNA or lead to indirect formation of DNA damage due to oxidative stress, which ultimately leads to single DNA strand breaks but no cross-linking of DNA. The oxidative stress caused by depletion of GSH due to reaction with high concentrations of ,s-unsaturated aldehydes may also lead subsequent release of nucleocytolytic enzymes, causing DNA fragmentation, cellular damage, and apoptosis. However, in vivo, there is no evidence of mutagenicity and genotoxicity in the mouse micronucleus test for 2-hexenal, 2-octenal, and 2-decenal, and no indication of unscheduled DNA synthesis for 2-hexenal in the liver (Adams et al., 2008). In addition, there is no evidence of DNA adduct formation in the liver of animals exposed to high dose levels (200 and 500 mg/kg body weight) of 2-hexenal for up to 28 days (Stout et al., 2008) and no evidence of hepatocarcinogenicity in rats or mice exposed to high dose levels of 2,4-hexadienal for up to 2 years (NTP, 2003). Based on these observations, the fact that normal human consumption of ,s-unsaturated aldehydes is many orders of magnitude less than those showing no effects in animals, and the rapid metabolism of these aldehydes in the high-capacity s-oxidation pathway in humans, it is concluded that use of the substances are GRAS under intended conditions of intake as flavor ingredients.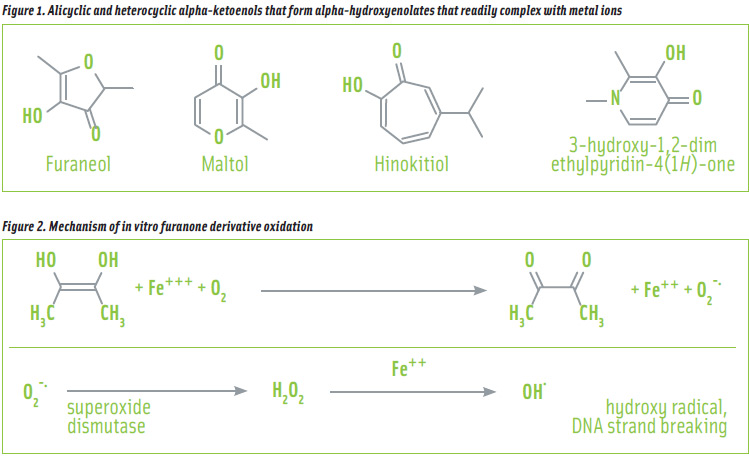
The other group of structurally related substances that shows evidence of genotoxicity in both in vitro and in some in vivo genotoxicity assays is alicyclic or heterocyclic alpha-ketoenols. These substances induce DNA damage in vitro by generating free radicals that induce strand scission (JECFA, 2006). In order to produce these cytotoxic eff ects, the structures must contain specific structural features. The alpha-ketoenol moiety must be present to bind the metal ion (Fe3+ or Cu2+) and the molecule must have a source of electrons from an electron donating group (e.g., an alkyl ring substituent or a heteroatom in the ring). Hence, similar biochemical behaviour has been reported for furaneol (4-hydroxy-2,5-dimethyl2(3H)-furanone) (Murakami et al., 2007); maltol (3-hydroxy-2-methyl-4-pyrone) (Murakami et al., 2005a, 2005b, 2006); hinokitiol (2-hydroxy-4-isopropyl-2,4,6-cycloheptatrienone) (Murakami et al. 2005a); and 3-hydroxy-1,2-imethylpyridin-4(1H)-one (Moridani and O’Brien, 2001) (see Figure 1). It has been reported that metal chelating ability does not strictly correlate to metal ion reduction ability (Murakami et al., 2007). The donation of electrons, either from an alkyl substituent or the ring itself allows the alpha-ketoenol to act as an alpha-hydroxyenolate that readily complexes with metal ions and provides an electron for metal reduction.
In one pathway, the alpha-ketoenol reduces the metal ion, which subsequently reduces molecular oxygen to produce superoxide ion. Superoxide dismutase catalyzes the conversion of superoxide ion to hydrogen peroxide that is then converted to hydroxyl radicals by Fe2+ in the Fenton reaction (see Figure 2). Hydrogen peroxide also oxidizes glutathione (GSH), leading to a decreased GSH/GSSG ratio and an increase in cellular oxidative stress.
Support for this pathway has been reported when various cell lines have been incubated with furaneol (Hiramoto, 1998), maltol (Murakami, 2005b), and hinokitiol (Ido et al., 2002; Inamori et al., 1993). In these experiments, the ketoenol/ iron complex led to inactivation of aconitase, an iron-sulfur cluster-containing enzyme with high sensitivity to reactive oxygen species. The inactivation of aconitase was metal ion-dependent, and prevented by 4-hydroxy-2,2,6,6-tetramethylpiperidinyloxy (TEMPOL), a scavenger of reactive oxygen species and superoxide dismutase, suggesting that the ketoenol/iron-mediated generation of superoxide anion is responsible for the inactivation of aconitase. Addition of furaneol or maltol to calf thymus DNA stimulated the formation of 8-hydroxy-2’-deoxyguanosine (8-OHdG), an indicator for the presence of hydroxyl radicals, and addition of catalase, an enzyme that converts hydrogen peroxide, a hydroxyl radical precursor, to water and oxygen, completely inhibits formation of 8-OHdG (Murakami, 2005b, 2007).
In one experiment, hinokitiol induced DNA fragmentation in teratocarcinoma F9 cells in a concentration and time dependent manner. In contrast, hinokitiol methyl ether and hinokitiol-Fe3+complex showed no DNA fragmentation in cells in culture or in cell-free systems. Hinokitiol activated caspase-3, one of the key enzymes in the apoptotic cascade (Ido et al., 2002). In conclusion, evidence of DNA reactivity in these genotoxicity assays is the result of reduction of intracellular metal ions by high cellular concentrations of alpha-ketoenols eventually leading to production of OH radicals leading to DNA damage and eventually apoptosis.
Despite the fact that these substances exhibit genotoxicity, they do not produce carcinogenic eff ects in rodents. Three studies, one on ethyl maltol, one on furaneol and the other on a structurally related furanone, show no evidence of carcinogenicity at intake levels at least four orders of magnitude greater than the intake of these substances added as flavouring agents (Kelly and Bolte, 2003; Munday and Kirkby, 1973; Gralla et al., 1969). Furthermore, vitamin C, a furanone containing a dihydroxyenol moiety with a genotoxicity test profile similar to that of DMHF, does not demonstrate carcinogenicity (NRC, 1996). In humans, furaneol (Roscher et al., 1997) and maltol (Rennhard, 1971) are rapidly absorbed in the gastrointestinal tract, conjugated with glucuronic acid in the liver and excreted in the urine. Free furaneol is not detected in the blood of human volunteers to whom it is administered as a constituent of strawberries; its glucuronic acid conjugate is the principal urinary metabolite (Roscher et al., 1997). At low levels of intake as flavouring substances, it is highly unlikely that these substances would exhibit the genotoxic potential observed under in vitro conditions at much higher cellular concentrations or in vivo at severely toxiclevels (>oral LD50 levels). Based on the available data, it is highly unlikely that these substances pose any significant genotoxic risk to humans under conditions of use as flavouring agents.
--- PAGE BREAK ---
For the other chemical groups containing more than 2,000 substances, there is no consistent positive evidence of mutagenicity or genotoxicity in a specific in vitroassay or in a group of standard in vitro assays (bacterial reverse mutation [Ames], mouse lymphoma, sister chromatid exchange, and chromosomal aberration). In cases where raw data are available for the in vitro assays, positive evidence of mutations, clastogenicity, or genotoxicity occurs at concentrations in which there is measurable evidence of cytotoxicity. Positive evidence of mutagenicity or genotoxicity in a single in vitro assay is not confirmed upon repeat analysis or by standardized in vivo assays (e.g., mouse micronucleus, unscheduled DNA synthesis) using oral administration.
Among the standard battery of genotoxicity assays, the in vitro chromosome aberration test, the sister chromatid exchange assay,and the mouse lymphoma assay, all show higher incidences of positive evidence of genotoxicity. While all of the standardized assays have value in detecting relevant genotoxicity under experimental conditions, there is no supportive evidence for DNA reactivity for a considerable number of in vitro clastogens in the chromosomal aberration or sister chromatid exchange assays or forward mutations in the mouse lymphoma assay. In fact, the clastogenic response of cells when exposed to fl avoring substances is strongly correlated with cytotoxicity. This may invoke the need to alter the protocol for assessing clastogenicity in vitro. Decreasing the level of cytotoxicity (e.g., from 50% to 10–20%) at which clastogenic responses are still considered valid and lowering the upper limits of test substance concentration, irrespective of cytotoxicity, may help ensure sufficient reliability of future genotoxicity testing with mammalian cells in vitro. These recommendations have recently been proposed by the International Conference on Harmonisation (ICH) and in the Organization for Economic Co-Operation & Economic Development’s (OECD) guidelines to create a common ground of understanding for interpretation of in vitro (positive) test results (Kirkland et al., 2005, 2007a, 2007b; Meintieres and Marzin, 2004; Muller and Sufuni, 2000; Thybaud et al., 2007).
Safety Assessment of Isoeugenol (FEMA No. 2468)
Isoeugenol (CAS No. 97-54-1) is trans-4-hydroxy-3-methoxy-1-propenylbenzene. Together with a number of structurally related derivatives, isoeugenol occurs in a wide variety of plants and plant extracts, including savory basil, clove, mace, dill seed, and nutmeg. As a flavoring agent, it has been added to flavor formulations for nonalcoholic beverages, candy, and chewing gum. The annual reported volume of use as a flavor ingredient is 236 kg in the U.S. and 426 kg in Europe (Gavin et al., 2008; EFFA, 2005).
In a 2-year bioassay sponsored by National Toxicology Program (NTP, 2008), groups of 50 F344/N rats or B6C3F1 mice of both sexes were administered oral doses of 0, 75, 150, or 300 mg/kg body weight per day of isoeugenol by gavage, 5 days/week for 105 weeks. The NTP Subcommittee that reviewed the study concluded that:
“Under the conditions of these 2-year gavage studies, there was equivocal evidence of carcinogenic activity of isoeugenol in male F344/N rats based on increased incidences of rarely occurring thymoma and mammary gland carcinoma. There was no evidence of carcinogenic activity in female F344/N rats…. There was clear evidence of carcinogenic activity of isoeugenol in male B6C3F1 mice based on increased incidences of hepatocellular adenoma, hepatocellular carcinoma, and hepatocellular adenoma or carcinoma (combined). There was equivocal evidence of carcinogenic activity of isoeugenol in female B6C3F1 mice based on increased incidences of histiocytic sarcoma.”
The primary neoplastic effects reported in the 2-year NTP gavage study were associated with the liver of control and treated male mice. The high incidence of hepatocellular adenomas, carcinomas, and adenomas and carcinomas (combined) in both control and treated groups of male mice is indicative of the sensitivity of the B6C3F1 male mouse liver to toxicity and eventually neoplastic changes. Although the incidence of hepatocellular adenomas (control, 24/50; dosed groups, 35/50 at 75 mg/kg bw, 37/50 at 150 mg/kg bw, and 33/50 at 300 mg/kg bw), hepatocellular carcinomas (control, 8/50; dosed groups, 18/50 at 75 mg/kg bw, 19/50 at 150 mg/kg bw, and 18/50 at 300 mg/kg bw), and combined hepatocellular adenomas and carcinomas (control, 28/50; dosed groups, 43/50 at 75 mg/kg bw, 43/50 at 150 mg/kg bw, and 43/50 at 300 mg/kg bw) in all groups of treated male mice was greater than in the control group, the effects were not dose-dependent and the incidence of malignant neoplasms (14/50) of all types (hepatocellular carcinomas, hemangiosarcoma, hepatocholangiocarcinomas, and hepatoblastomas) in the control group was similar to the incidences in the treated groups. No liver neoplastic effects were reported in rats.
This pattern of neoplastic responses is consistent with the historically high levels of background hepatocellular neoplasms in male B6C3F1 mice (Maronpot et al., 1987). The historical spontaneous incidence of liver neoplasms in control male B6C3F1 mice has revealed background incidences of combined hepatocellular adenoma and carcinomas of 32.4% for males with a range of 20–47% (NTP, 2006). A higher incidence was reported in the control group of the isoeugenol study (56%). It is generally well accepted that male and female B6C3F1 mouse liver tumors that arise in 2-year bioassays with various agents (e.g., chloroform) are an indirect result of dose-related chronic toxicity and resulting cellular proliferation. In the absence of this chronic toxicity in humans, these tumors are not considered to represent a risk for humans (Cohen et al., 2004).
There is substantial evidence that the appearance of male B6C3F1 mouse liver tumors is not relevant to a human risk. First, there was no statistical evidence of an increased incidence of hepatic tumors in female mice or male or female rats related to administration of isoeugenol. Second, all dose groups of male B6C3F1 mice suff ered chronic hepatic toxicity prior to the development of either liver adenomas or carcinomas, as evidenced by the results of the 90-day and 2-year studies. Hepatocellular adenomas and carcinomas also occurred late in the life span of males. From a biological perspective, the increase in the incidence of tumors in male B6C3F1 mice refl ects the impact of high-dose liver damage to an organ already prone to spontaneous development of liver neoplasms (Haseman et al., 1986, 1990).
Therefore, it can be concluded that the carcinogenic potential in this sensitive breed and sex of laboratory rodent is a secondary biological response to dose-dependent hepatotoxicity, and is not relevant to humans who consume isoeugenol at low non-toxic levels (<0.1 mg kg bw per day) from intended use as a flavoring ingredient. these levels of intake are at least six orders of magnitude less than those used in the ntp study.>
--- PAGE BREAK ---
The only other neoplastic eff ect reported was in female mice. There was an increased incidence of histiocytic sarcoma in dosed compared to control females (control, 0/50; dosed groups, 1/50 at 75 mg/kg bw; 1/50 at 150 mg/kg bw; 4/50 at 300 mg/kg bw.) Although no evidence of histiocytic sarcoma was reported in control females, the incidence of histiocytic sarcomas (1.5% } 2.2%, range 0–8%) and incidences of tumors in the isoeugenol study (2% and 8%) are well within the historical control range for the NTP female mouse historical database.
In order to evaluate the relevance of these tumors to the potential cancer risk for humans consuming isoeugenol as a flavoring agent, it is important to note that a significant increase in histiocytic sarcomas was reported at dose levels inducing liver toxicity and even necrosis (8/50 at 300 mg/kg bw per day). Increased incidences of splenic extramedullary hematopoiesis and splenic cellular depletion are additional evidence of damage to the hematopoietic system.
Clearly, low incidences of histiocytic sarcomas occurred in only one sex, and only at dose levels inducing toxicity, rendering it highly likely that the occurrence of these tumors is a high-dose phenomenon that manifests itself in severely affected livers of female mice. The estimated daily per capita intake of isoeugenol as a flavoring agent in the U.S. (0.00002 mg/kg bw/day) is more than 3,750,000 times lower than the dose levels in the NTP study. Therefore, the occurrence of these neoplasms in the present study is considered a high-dose phenomenon without any relevance for assessing the potential cancer risk of the use of isoeugenol as a food flavor ingredient.
VIEW GRAPHS
Download the PDF Version of this article to view GRAS tables and graphs.
FEMA GRAS LISTS published in Food Technology, in chronological order
Hall, R.L. 1960. Recent progress in the consideration of fl avoring ingredients under the Food Additives Amendment. Food Technol. 14: 488-495.
Hall, L. and Oser, B.L. 1961. Recent progress in the consideration of flavoring ingredients under the Food Additives Amendment. II. Food Technol. 15(12): 20, 22-26.
Hall, R.L. and Oser, B.L. 1965. Recent progress in the consideration of flavoring ingredients under the Food Additives Amendment. 3. GRAS substances. Food Technol. 19(2, Part 2): 151-197.
Hall, R.L. and Oser, B.L. 1970. Recent progress in the consideration of flavoring ingredients under the Food Additives Amendment. 4. GRAS substances. Food Technol. 24(5): 25-34.
Oser, B.L. and Hall, R.L. 1972. Recent progress in the consideration of flavoring ingredients under the Food Additives Amendment. 5. GRAS substances. Food Technol. 26(5): 35-42.
Oser, B.L. and Ford, R.A. 1973a. Recent progressin the consideration of flavoring ingredients under the Food Additives Amendment. 6. GRAS substances. Food Technol. 27(1): 64-67.
Oser, B.L. and Ford, R.A. 1973b. Recent progress in the consideration of flavoring ingredients under the Food Additives Amendment. 7. GRAS substances. Food Technol. 27(11): 56-57.
Oser, B.L. and Ford, R.A. 1974. Recent progress in the consideration of flavoring ingredients under the Food Additives Amendment. 8. GRAS substances. Food Technol. 28(9): 76-80.
Oser, B.L. and Ford, R.A. 1975. Recent progress in the consideration of flavoring ingredients under the Food Additives Amendment. 9. GRAS substances. Food Technol. 29(8): 70-72.
Oser, B.L. and Ford, R.A. 1977. Recent progress in the consideration of flavoring ingredients under the Food Additives Amendment. 10. GRAS substances. Food Technol. 31(1): 65-74.
Oser, B.L. and Ford, R.A. 1978. Recent progress in the consideration of flavoring ingredients under the Food Additives Amendment. 11. GRAS substances. Food Technol. 32(2): 60-70.
Oser, B.L. and Ford, R.A. 1979. Recent progress in the consideration of flavoring ingredients under the Food Additives Amendment. 12. GRAS substances. Food Technol. 33(7): 65-73.
Oser, B.L., Ford, R.A., and Bernard, B.K. 1984. Recent progress in the consideration of flavoring ingredients under the Food Additives Amendment. 13. GRAS substances. Food Technol. 38(10): 66-89.
Oser, B.L., Weil, C.L., Woods, L.A., and Bernard, B.K. 1985. Recent progress in the consideration of flavoring ingredients under the Food Additives Amendment. 14. GRAS substances. Food Technol. 39(11): 108-117.
Burdock, G.A., Wagner, B.M., Smith, R.L., Munro, I.C., and Newberne, P.M. 1990. Recent progress in the consideration of flavoring ingredients under the Food Additives Amendment. 15. GRAS substances. Food Technol. 44(2): 78, 80, 82, 84, 86.
Smith, R.L. and Ford, R.A. 1993. Recent progress in the consideration of flavoring ingredients under the Food Additives Amendment. 16. GRAS substances. Food Technol. 47(6): 104-117.
Smith, R.L., Newberne, P., Adams, T.B., Ford, R.A., Hallagan, J.B., and the FEMA Expert Panel. 1996a. GRAS flavoring substances 17. Food Technol. 50(10): 72-78, 80-81.
Smith, R.L., Newberne, P., Adams, T.B., Ford, R.A., Hallagan, J.B., and the FEMA Expert Panel. 1996b. Correction to GRAS flavoring substances 17. Food Technol. 51(2): 32.
Newberne, P., Smith, R.L., Doull, J., Goodman, J.I., Munro, I.C., Portoghese, P.S., Wagner, B.M., Weil, C.S., Woods, L.A., Adams, T.B., Hallagan, J.B., and Ford, R.A. 1998. GRAS flavoring substances 18. Food Technol. 52(9): 65-66, 68, 70, 72, 74, 76, 79- 92.
Newberne, P., Smith, R.L., Doull, J., Goodman, J.I., Munro, I.C., Portoghese, P.S., Wagner, B.M., Weil, C.S., Woods, L.A., Adams, T.B., Hallagan, J.B., and Ford, R.A. 1999. Correction to GRAS flavoring substances 18. Food Technol. 53(3): 104.
Newberne, P., Smith, R.L., Doull, J., Feron, V.J., Goodman, J.I., Munro, I.C., Portoghese, P.S., Waddell, W.J., Wagner, B.M., Weil, C.S., Adams, T.B., and Hallagan, J.B. 2000. GRAS flavoring substances 19. Food Technol. 54(6): 66, 68-69, 70, 72-74, 76-84.
Smith, R.L., Doull, J., Feron, V.J., Goodman, J.I., Munro, I.C., Newberne, P.M., Portoghese, P.S., Waddell, W.J., Wagner, B.M., Adams, T.B., and McGowen, M.M. 2001. GRAS flavoring substances 20. Food Technol. 55(12): 34-36, 38, 40, 42, 44-55.
Smith, R.L., Cohen, S.M., Doull, J., Feron, V.J., Goodman, J.I., Marnett, I.J., Portoghese, P.S., Waddell, W.J., Wagner, B.M., and Adams, T.B. 2003. GRAS flavoring substances 21. Food Technol. 57(5): 46-48, 50, 52-54, 56-59.
Smith, R.L., Cohen, S.M., Doull, J., Feron, V.J., Goodman, J.I., Marnett, I.J., Portoghese, P.S., Waddell, W.J., Wagner, B.M., and Adams, T.B. 2005. GRAS flavoring substances 22. Food Technol. 59(8): 24-28, 31-32, 34, 36-62.
Waddell, W.J., Cohen, S.M., Feron, V.J., Goodman, J.I., Marnett, L.J., Portoghese, P.S., Rietjens, I.M.C.M., Smith, R.L., Adams, T.B., Gavin, C. L., McGowen, M.M., and Williams, M.C. 2007. GRAS flavoring substances 23. Food Technol. 61(8): 22-24, 26-28, 30-49.
CORRECTION & CHANGES
• Alternate Citronella Source. Citronella oil (FEMA 2308) reported in GRAS 3 (Hall and Oser, 1965) incorrectly listed only one species, Cymbopogon nardus Rendle, as the accepted botanical source. An alternate species, Cymbopogon winterianus Jowitt, is also an accepted botanical source for GRAS approved citronella oil.
• DeGRAS of Acetamide. The substance acetamide (FEMA No. 4251) was reviewed by the Expert Panel and found to require additional data, including chronic toxicity testing, for the continuation of its GRAS status. Based on a survey of the flavor industry, there was insuffi cient interest to support the acquisition of the requested data. Therefore, the flavor ingredient acetamide has been removed from the GRAS list.
• Expert Panel Member Change. Nigel Gooderham, Professor of Pharmacology and Toxicology at University of London, joined the Panel in January 2009.
• Consultant Change. Ian Munro, a Panel member from 1983 to 2001 and a Panel consultant beginning in 2001, retired from the Panel in October 2008. He has spent more than a quarter of a century in service to the Expert Panel and the fl avor industry. His experience in toxicology, international food and flavor safety, and food and flavor regulation provided the Panel with a unique expertise that contributed significantly to successful operation of the Panel.
References
Adams, T.B., Cohen, S.M., Doull, J., Feron, V.J., Goodman, J.I., Marnett, L.J., Munro, I.C., Portoghese, P.S., Smith, R.L., Waddell, W.J., and Wagner, B.M. 2005. The FEMA GRAS assessment of benzyl derivatives used as fl avor ingredients. Food Chem. Toxicol. 43: 1207-1240.
Adams, T.B., Gavin, C.L., Taylor, S.V., Waddell, W.J., Cohen, S.M., Feron, V.J., Goodman, J.I., Rietjens, I.M.C.M., Marnett, L.J., Portoghese, P.S., and Smith, R.L. 2008. The FEMA GRAS assessment of alpha,beta-unsaturated aldehydes and related substances used as fl avor ingredients. Food Chem. Toxicol. 49: 2935-2967.
Cohen, S.M., Klaunig J., Meek, M.E., Hill, R.N., Pastoor, T., Lehman-McKeeman, L., Bucher, J., Longfellow, D.G., Seed, J., and Dellarco, V. 2004. Evaluating the human relevance of chemically induced animal tumors. Toxicol. Sci. 78: 181-186.
EFFA. 2005. European Flavour and Fragrance Association European inquiry on volume use. Private communication to the Flavor and ExtractManufacturers Association (FEMA).
Gavin, C.L., Williams, M.C., and Hallagan, J.B. 2008. FEMA 2005 poundage and technical eff ects update survey. Flavor and Extract Manufacturers Association, Washington, D.C.
Gralla, E.J., Stebbins, R.B., Coleman G.L., and Delahunt, C.S. 1969. Toxicity studies with ethyl maltol. Toxicol. App. Pharmacol. 15(3): 604-613.
Hall, R.L. and Oser, B.L. 1965. Recent progress in the consideration of fl avor ingredients under the Food Additives Amendment. 3 GRAS Substances. Food Technol. 19(2, Part 2): 151-197.
Haseman, J.K., Winbush, J.S., and Ob�0;0;0;0;0;19;Donnell, Jr., M.W. 1986. Use of dual control groups to estimate false positive rates in laboratory animal carcinogenicity studies. Fundamental and Applied Toxicol. 7: 573-584.
Haseman, J.K., Zeiger, E., Shelby, M.D., Margolin, B.H., and Tennant, R.W. 1990. Predicting rodent carcinogenicity from four in vitro genetic toxicity assays: an evaluation of 114 chemicals studied by the National Toxicology Program. J. Am. Statistical Assoc. 85(412): 964-971.
Hiramoto, K., Kato, T., Takahashi, Y., Yugi, K., and Kikugawa, K. 1998. Absorption and induction of micronucleated peripheral reticulocytes in mice after oral administration of fragrant hydroxyfuranones generated in the Maillard reaction. Mutat. Res. 415: 79-83.
Ido, Y., Muto, N., Inada, A., Kohroki, J., Mano, M., Odani, T., Itoh, N., Yamamoto, K., and Tanaka, K. 2002. Induction of apoptosis by hinokitiol, a potent iron chelator, in teratocarcinoma F9 cells is mediated through the activation of caspase-3. Cell Proliferation 32(1): 63-73.
Inamori, Y., Tsujibo, H., Ohishi, H., Ishii, F., Mizugaki, M., Aso, H., and Ishida, N. 1993. Cytotoxic eff ect of hinokitiol and tropolone on the growth of mammalian cells and on blastogenesis of mouse splenic T cells. Biol. Pharm. Sci. 16(5): 521-523.
JECFA. 2006. Safety evaluation of certain food additives. Report of the Joint FAO/WHO Expert Committee on Food Additives. WHO Food Additives Series No. 54. World Health Org., Geneva.
Kelly, C.M. and Bolte, H.F. 2003. ST 07 C99: a 24-month dietary carcinogenicity study in rats. Final report. Study No. 99-2644. Huntingdon Life Sciences, East Millstone N.J. Private communication to the Flavor and Extract Manufacturers Association.
Kirkland, D., Aardema, M., Henderson., L., and Muller, L. 2005. Evaluation of the ability of a battery of three in vitro genotoxicity tests to discriminate rodent carcinogens and non-carcinogens I. Sensitivity, specifi city, and relative predictivity. Mutat. Res. 584(1-2): 1-256.
Kirkland, D., Pfuhler, S., Tweats, D., Aardema, M., Corvi, R., Darroudi, F., Elhajouji, A., Glatt, H., Hastwell, P., Hayashi, M., Kasper, P., Kirchner, S., Lynch, A., Marzin, D., Maurici, D., Meunier, J.R., Muller, L., Nohynek, G., Parry, J., Parry, E., Thybaud, V., Tice, R., van Benthem, J., Vanparys, P., and White, P. 2007a. How to reduce false positive results when undertaking in vitro genotoxicity testing and thus avoid unnecessary followup animal tests: Report of an ECVAM Workshop. Mutat. Res. 628(1): 31-55.
Kirkland, D., Aardema, M., Banduhn, N., Carmichael, P., Fautz, R., Meunier, J., and Pfuhler, S. 2007b. In vitro approaches to develop weight of evidence (WoE) and mode of action (MoA) discussions with positive in vitro genotoxicity results. Mutagenesis 22(3): 161-175.
Maronpot, R.R., Haseman, J.K., Boorman, G.A., Eustis, S.E., Rao, G.N., and Huff , J.E. 1987. Liver lesions in B6C3F1 mice: the National Toxicology Program, experience and position. Arch. Toxicol. 10: 10-26.
Meintieres, S. and Marzin, D. 2004. Apoptosis may contribute to false-positive results in the in vitro micronucleus test performed in extreme osmolality, ionic strength, and pH conditions. Mutat. Res. 560: 101-118.
Moridani, M.Y. and Ob�0;0;0;0;0;19;Brien, P.J. 2001. Ion complexes of deferiprone and dietary plant catechols as cytoprotective superoxide radical scavengers. Biochem. Pharmacol. 62: 1579-1585.
Muller, L. and Sofuni, T. 2000. Appropriate levels of cytotoxicity for genotoxicity tests using mammalian cells in vitro. Environ. Mol. Mutagen. 35: 202-205.
Munday, R. and Kirkby, W. 1973. Unpublished report. Biological evaluation of a fl avor cocktail. III. One-year feeding study in rats. Private communication to FEMA, Washington, D.C.
Murakami, K., Ohara, Y., Haneda, M., Tsubouchi, R., and Yoshino, M. 2005a. Prooxidant Action of Hinokitiol: Hinokitiol-Iron Dependent Generation of Reactive Oxygen Species. Basic Clin. Pharmacol. Toxicol. 97(6): 392-394.
Murakami, K., Ishida, K., Watakabe, K., Tsubouchi, R., Haneda, M., and Yoshino M. 2005b. Prooxidant action of maltol: Role of transition metals in the generation of reactive oxygen species and enhanced formation of 8-hydroxy-2b�0;0;0;0;0;19;-deoxyguanosine formation in DNA. BioMetals 19: 253-257.
Murakami, K., Ishida, K., Watakabe, K., Tsubouchi, R., Naruse, M., and Yoshino, M. 2006. Maltol/iron-mediated apoptosis in HL60 cells: Participation of reactive oxygen species. Toxicol. Lett. 161(2): 102-107.
Murakami, K., Haneda, M., Makino, T., and Yoshino, M. 2007. Prooxidant action of furanone compounds: Implication of reactive oxygen species in the metaldependent strand breaks and the formation of 8-hydroxy-2b�0;0;0;0;0;19;-deoxyguanosine in DNA. Food Chem. Toxicol. 45: 1258-1262.
NRC. 1996. Carcinogens and anticarcinogens in the human diet: a comparison of naturally occurring and synthetic substances. National Academy Press, 91-92. National Research Council, Washington, D.C.
NTP. 2003. Technical Report on the Toxicology and Carcinogenesis Studies of 2,4-Hexadienal (CAS No. 142-83-6) in F344/N Rats and B6C3F1 Mice. NTP TR 509. NIH Publication No. 04-4443. Natl. Toxicology Program, Research Triangle Park, N.C. http://ntp.niehs.nih.gov/
NTP. 2006. Technical Report on the Toxicology and Carcinogenesis Studies of Benzophenone (CAS NO. 119-61-9) in F344/N Rats and B6C3F1 Mice. NTP TR 533. NIH Publication No. 06-4469. Natl. Toxicology Program, Research Triangle Park, N.C. http://ntp.niehs.nih.gov/
NTP. 2008. Draft Report: Technical Report on the Toxicology and Carcinogenesis Studies of Isoeugenol (CAS NO. 97-54-1) in F344/N Rats and B6C3F1 Mice. NTP TR 551. Natl. Toxicology Program, Research Triangle Park, N.C.
Rennhard, H.H. 1971. The metabolism of ethyl maltol and maltol in the dog. J. Agric. Food Chem. 19(1): 152-154.
Roscher, R., Koch, H., Herderich, M., Schreier, P., and Schwab, W. 1997. Identifi cation of 2,5-Dimethyl-4-hydroxy- 3[2H]-franone beta-D-Glucuronide as the Major Metabolite of Strawberry Flavour Constituent in Humans. Food Chem. Toxicol. 35: 777-782.
Stout, M., Bodes, E., Schoonhoven, R., Upton, P., Travlos, G., and Swenberg, J. 2008. Toxicity, DNA binding, and cell proliferation in male F344 rats following short-term gavage exposures to trans-2hexenal. Toxicology Pathology 36(2): 232-246.
Thybaud, V., Aardema, M., Clements, J., Dearfi eld, K., Galloway, S., Hayashi, M., Jacobson-Kram, D., Kirkland, D., MacGregor, J., Marzin, D., Ohyama, W., Schuler, M., Suzuki, H., and Zeiger, E. 2007. Strategy for genotoxicity testing: Hazard identifi cation and risk assessment in relation to in vitro testing. Mutat. Res. 627: 41-58.
Authors
-
M.C. WILLIAMS
Categories
-
Food Health Nutrition
-
Food Safety and Defense
-
Food Ingredients and Additives
-
Food Policy
-
Colors
-
Food Technology Magazine



