Peak Protein: Today’s Innovations, Tomorrow’s Transformations

-
Learning Objective 1
Understand the current and future educational and workforce needs to enable the commercial success of the emerging fermentation, fermentation-derived, and cultivated alternative protein and functional food ingredients industry.
-
Learning Objective 2
Obtain a new perspective on how microbiology and traditional fermentation, biomass fermentation, precision fermentation, and cultivated meat/seafood could positively impact reaching many of the UN’s SDG goals.
-
Learning Objective 3
Discover which enabling technologies will fuel the future transformation of our global food supply, ensuring food security for all while protecting our planet.
As discussed in Part 1 of this series, fermentation is one of the earliest forms of food processing and of biotechnology. As omics, systems biology, and biotechnology techniques and processes have advanced, precision fermentation (PF) is the next evolution of this ancient process. PF focuses on utilizing microbial host cells (e.g., yeasts, fungi, mycelia, and microalgae) as “factories” to produce specific molecules using bioreactors and fermentation processes. The host cells are typically genetically engineered but can be native strains that produce specific molecules. Following the fermentation process, typically using submerged, solid state, or membrane cell-recycle bioreactors, the microbial cells (waste stream) are separated from the fermentation medium and the target molecule is concentrated and purified.
One of the earliest examples of PF is the commercial production of human insulin in 1982 using genetically engineered (recombinant) Escherichia coli (E. coli). The first commercial food ingredient was chymosin B, also produced using recombinant E. coli as the host strain. It was approved and given generally recognized as safe (GRAS) status by the U.S. Food and Drug Administration (FDA) in 1990.
There are dozens of natural functional food ingredients produced today via PF. As future targets are broad, including lipids, fatty acids, proteins, flavors, enzymes, growth factors, pigments, etc., the possibilities for any food ingredient to be produced at scale in the future are unlimited. There has been an explosion of innovation and investment in startups in the alternative protein space. PF-derived product examples include development and/or commercialization of albumen and other egg proteins, pepsin, collagen, elastin, whey, casein, colors, soy leghemoglobin protein (a substitute for hemoglobin), heme proteins, cell growth factors (for cultivated meat culture media), and many other animal-free ingredients that can help deliver a dairy/meat/egg/poultry-like experience in alternative proteins.
FDA regulates these products via the GRAS notification process, either self-affirmed or optional notified GRAS. Additionally, if no viable genetically modified (GM) production host cells or DNA is in the product, it does not need to be labeled as a bioengineered food. If GM host strain DNA is present in the final product, it must be labeled as a bioengineered food. The European Food Safety Authority regulates these products in the European Union (EU) via novel foods and transparency processes. If no viable GM production host cells or DNA are present in the final product, they are regulated as precision fermentation food with no special labeling. If the GM host is not removed from the product, it will fall under the scope of the EU’s GM food regulations and directives, authorized and labeled accordingly.
Cultivated Protein and Fat
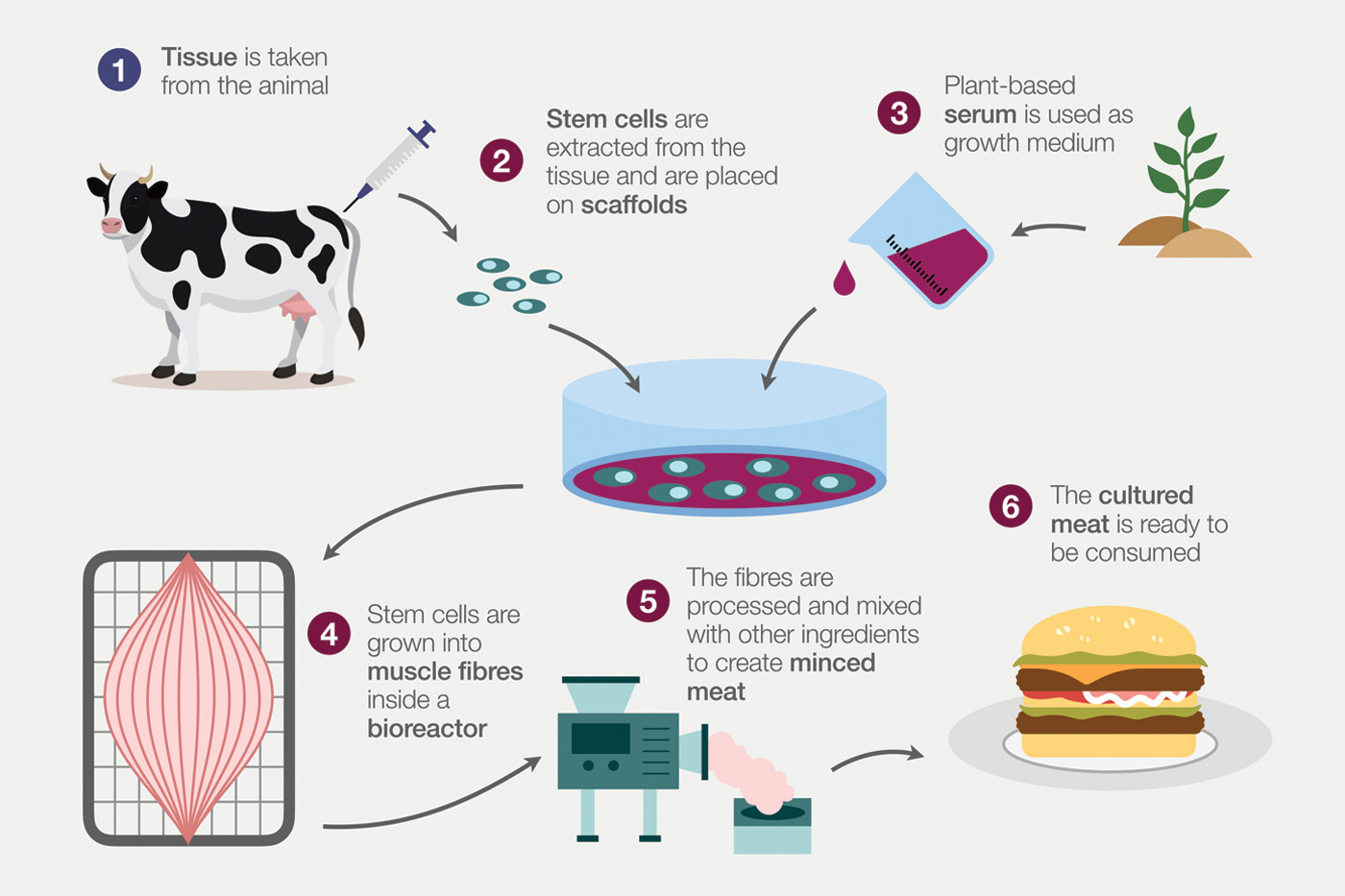
Cell culture is the process in which animal or plant cells are grown outside the body or plant under controlled conditions. Significant development occurred in the 1940s and 50s, and the first commercial product was the polio vaccine. Today, a wide variety of pharmaceutical products are produced via animal cell culture in a process similar to PF, utilizing host cells for production of a targeted molecule. Cultivated meat (CM) is similar to biomass fermentation (BF), where the focus is on producing large quantities of animal cells and/or tissue (e.g., muscle and fat) while minimizing production of expressed metabolites. Therefore, it requires much larger scale bioreactors than those commercially available and used by the pharmaceutical industry. This is a critical hurdle in commercialization of CM products.
CM is real animal meat (including poultry and seafood) produced by cultivating animal cells directly in bioreactors. The process involves biopsies from the animal to develop master cell lines that are isolated, purity ensured, and stored frozen. From the frozen stock, seed cultures are prepared in small bioreactors and transferred to inoculate large bioreactors (typically stirred tank or airlift) for cell proliferation and differentiation. The cells/tissue are harvested from the bioreactor. Downstream processing may include food formulation and/or processing, packaging, and storage prior to distribution.
In the United States, FDA regulates CM production from the development of the master cell line through production in bioreactors and harvest of the CM cells/tissue. The U.S. Department of Agriculture (USDA) takes responsibility from harvest up to and including production of final CM products. In 2023, two companies—Upside Foods and GOOD Meat—received the first U.S. regulatory approval for their cultivated chicken products. Singapore and Israel have approved CM products for human consumption. In the EU, CM is governed by the novel food regulation. Many governments are acting on the sustainability and food security benefits of alternative proteins, using public funds to support both CM research and development (R&D) and startups.
The bio-based economy is expected to grow by at least 50% by 2030.
Next-Generation Talent
According to a 2021 Global Innovation Needs Assessment by ClimateWorks Foundation, the alternative protein market “could create more than 8 million jobs globally by 2040, and up to 9.5 million by 2050.” There are almost 2,000 alternative protein companies across the value chain, creating thousands of new jobs worldwide. Critical needs include engineers with a bioprocess or regulatory specialization and scientists from adjacent fields, including stem cell biology, materials science, protein chemistry, food science, and plant science (Good Food Institute 2023).
The Good Food Institute (GFI) surveyed its global community of alternative protein startups on workforce needs. The results included demand for:
- Scientific and engineering capacity for R&D roles and operators and technicians in manufacturing, quality assurance, and quality control.
- Cross-production platform skills in disciplines including food science, meat science, process development and scale-up, manufacturing, product development, and food safety.
- Downstream process engineers who are skilled in PF and BF to support scaling up and PF for microbial strain development.
- In the area of CM, there is a need for bioreactor operators/controllers, scaffolding design experts, cell culture media developers, cell line engineers, and tissue engineering experts to support research and scale-up.
- Universities and professional associations are needed to ensure their members and students understand career opportunities and can obtain the education, training, and skills needed.

United Nations Sustainable Development Goals. (Note: The content of this publication has not been approved by the United Nations and does not reflect the views of the United Nations or its officials or member states.) Credit: Reprinted per usage permissions of the United Nations Sustainable Development Goals website (https://www.un.org/sustainabledevelopment).
Impact on SDGs
In 2015, the 2030 Agenda for Sustainable Development was adopted by all United Nations member states. It provides a shared blueprint for peace and prosperity for people and the planet, now, and into the future (United Nations 2015). At its heart are 17 Sustainable Development Goals (SDGs), several of which involve the production and sustainability of protein-rich foods on a global scale. In recent years, researchers have described how to accelerate progress in achieving the SDGs through microbiology (Fagunwa and Olanbiwoninu 2020). Similarly, traditional fermentation, BF, PF, and CM could help reach some SDG goals:
- SDG 2: Zero Hunger. Fermented foods already provide a significant portion of the diet in many cultures and are key in ensuring access to safe and nutritious food. Production of food and food ingredients via BF, PF, and CM will provide sustainable, additional sources of food to help ensure food security.
- SDG 3: Good Health and Well-Being. Probiotics and fermented foods have many known health benefits. PF can provide a variety of functional, nutritious ingredients, and along with CM, can provide healthy alternatives to traditional animal-based foods.
- SDGs 8 and 9: Decent Work and Economic Growth; and Industry, Innovation, and Infrastructure. The bio-based economy is expected to grow by at least 50% by 2030. As biotechnology has an exceptionally broad diversity of products and applications, it is capable of creating decent jobs and sustaining economic growth throughout the global food industry.
- SDG 12: Responsible Consumption and Production. Shelf-life extension via traditional fermentation (TF) supports production sustainability efforts that reduce food loss and waste via natural preservation.
- SDG 13: Climate Action. Alternative protein production can significantly reduce greenhouse gas emissions from the food system. Less land usage can free up land for production of renewable energy and preservation of biodiversity.
- SDG 14: Life Below Water. Alternative proteins produced via TF, BF, PF, and CM can reduce agricultural land use and inputs, limit water withdrawals, improve water quality, and reduce ocean pollution and degradation from agricultural runoff. It can take pressure off wild fisheries and aquaculture systems while meeting the increasing global demand for seafood (Good Food Institute n.d.).
- SDG 15: Life on Land. Opportunity costs for land use (if not used to grow crops or raise livestock) could be recouped by regrowing forests or wild grasslands, allowing us to take some of the 1,400 billion tons of CO2back out of the atmosphere and put it back into vegetation (Richie 2021). Land spared can reduce deforestation and protect biodiversity.
- SDG 17: Partnerships for the Goals. More partnerships promoted within and across countries are critical to establish public-private-academic and other partnerships to enable R&D and commercial scale-up of all fermentation and CM production of foods and functional ingredients.
The alternative protein market could create more than 8 million jobs globally by 2040, and up to 9.5 million by 2050.
Future Proofing the Food System
There are several challenges to overcome in the broader industrialization and commercialization of biotechnologies such as TF, BF, PF, and CM. These include consumer acceptance, food regulation, achieving market acceptable cost, taste, and scale-up and commercial infrastructure capacities. With continued public and private investment and innovation, these challenges can be met.
Each fermentation and cell culture method has the power to make protein and functional food ingredient production more efficient. These proteins and ingredients can complement each other, allowing companies to create more environmentally sustainable and less resource-intensive products (Good Food Institute 2022). This approach also opens broader opportunities to develop not only protein products that mimic their animal-based counterparts, but also to enable innovation of unique foods and functional ingredients that can deliver against ever evolving global consumers’ needs and desires.
© Dimitrios Karamitros/E+/Getty Images Plus
Today, the future transformation of our global food production system is being enabled by many exciting advances in science and technology. Industry 4.0 technologies such as artificial intelligence and machine learning, Internet of Things sensors and devices, and robotics are increasing interconnection and high-level automation to achieve smart food processing through fermentation and cell culture systems. Technologies that spur the interdisciplinary nature of knowledge related to physical, digital, and biological domains (such as 3-D printing) will increase the speed of food innovation. The convergence of advances in genome-based technologies and traditional fermentation enables the continued evolution of fermentation to produce sustainable, nutritious proteins and functional ingredients. Science at the intersection of synthetic biology, fermentation, bioreactor design, downstream processing, and food science is gaining speed, supporting technological developments to produce fermentation-derived and cultivated food and ingredients.
Now we’re ready to go back and consider the original question posed here: Does the key to meeting global protein demands and sustainable food production lie in the microscopic worlds of fermentation and cellular agriculture? With continued investment and innovation and keeping the SDGs and ethics in mind, there is unimaginable potential for these technologies to revolutionize our global food system, ensuring food security for all while protecting our planet.ft
To learn more
Does the key to meeting global protein demands and sustainable food production lie in the microscopic worlds of fermentation and cellular agriculture? In Part 1 of this two-part series, we first explored this question by diving into traditional fermentation and fermentation processes to produce biomass (Stewart 2024). In Part 2, we will explore the question through the lens of precision fermentation and cultivated proteins processes.
Hero Image: © guteksk7/E+/Getty Images Plus
Authors
-

Cindy Stewart Founder and Principal
Cindy Stewart, PhD, CFS, is the founder and principal of Innovative Food Science Consulting, which provides consulting, advisory, and technical due diligence services to the food and food ingredients, biotech/food tech, and venture capital industries. Stewart is past president (2017–2018) and Fellow of the Institute of Food Technologists.
Categories
-
Sustainability
-
Food Sciences
-
Fermentation
-
Biotechnology
-
Novel Technologies
-
Food Quality
-
Applied Science
-
Food Microbiology
-
Food Technology Magazine
-
Food Processing and Technologies



