Preserving Foods through High-Pressure Processing

Pressure treatment can be used to process both liquid and high-moisture-content solid foods. Although lethal to microorganisms, pressure treatment does not break covalent bonds and has a minimal effect on food chemistry. Thus, HPP provides a means for retaining food quality while avoiding the need for excessive thermal treatments or chemical preservatives.
High-pressure processing is also referred to as high-hydrostatic-pressure processing (HHP) or ultra-high-pressure processing (UHP) in the literature. Regardless of its nomenclature, the technology has been cited as one of the best innovations in food processing in 50 years (Dunne, 2005). The commercial use of high-pressure processing has been expanding. It provides food processors an opportunity to preserve foods with a “cleaner” ingredient label, and it is the process of choice for applications where heat pasteurization would adversely affect product quality. For example, in assuring the food safety of a product like salami, in-pack pasteurization using heat would result in liquid purge and fat migration. HPP allows products such as sliced deli meat to be pasteurized after slicing and packaging, thus mitigating the risk of contamination from the manufacturing environment. The U.S. Dept. of Agriculture, Food Safety and Inspection Service (USDA-FSIS) recognizes high-pressure processing as an acceptable food safety intervention for eliminating Listeria monocytogenes in processed meat products. Pressure treatment is also effective in inactivating other hazardous microorganisms such as E. coli, Salmonella, and Vibrio, as well as many yeasts, molds, and bacteria responsible for food spoilage. The microbiological shelf life and quality of foods can be substantially extended by the use of HPP (Hayman et al., 2004; He et al., 2002).
To date, HPP has only been used to inactivate vegetative microorganisms, but the use of high pressure in combination with heat to eliminate spores under conditions more favorable to quality than retorting is a topic of current research. Pathogenic spores such as Clostridium botulinum and varieties of Bacillus and Clostridia spoilage spores can potentially be eliminated through synergies of heat and pressure. This allows food to be sterilized with less thermal exposure (Matser et al., 2004; Margosch et al., 2006; Ahn et al., 2007). The quality of food produced using conditions of heat and pressure that are thought to lead to shelf stability in low-acid foods (105°C+, 700 MPa) is greatly improved over thermal processing (Lau and Turek, 2007). Reduced process times are an additional incentive.
Pressure treatment can also be used to alter the functional and sensory properties of various food components, especially proteins, allowing these to be beneficially altered (San Martín et al., 2002). Meat, fish, egg, and dairy proteins can be denatured with HPP in the absence of elevated temperatures. Increased viscosity and opacity are obtained with little change in fresh flavor. On the other hand, high pressure has very little effect on low-molecular-weight compounds such as flavor compounds, vitamins, and pigments compared to thermal processes. Accordingly, the quality of HPP pasteurized food is very similar to that of fresh food products. The quality throughout shelf life is influenced more by subsequent distribution and storage temperatures and the barrier properties of the packaging rather than by the pressure treatment. This article provides an overview on current technology status. Readers interested to learn more in depth can refer to extensive literature published on this subject (see References).
Basic HPP Principles
Various physical and chemical changes result from the application of pressure. Generally, physical compression during pressure treatment results in a volume reduction and an increase in temperature and energy (Heremans, 2003). In predicting the effect of HPP on foods, it is necessary to consider the net combined pressure-temperature effect of the process. The following principles govern the behavior of foods under pressure.
--- PAGE BREAK ---
• Le Chatelier’s principle. Any phenomenon (phase transition, change in molecular configuration, chemical reaction) accompanied by a decrease in volume is enhanced by pressure. Accordingly, pressure shifts the system to that of lowest volume (Farkas and Hoover, 2000).
• Principle of microscopic ordering. At constant temperature, an increase in pressure increases the degree of ordering of molecules of a given substance. Therefore, pressure and temperature exert antagonistic forces on molecular structure and chemical reactions (Balny and Masson, 1993).
• Isostatic principle. Pressure is uniformly distributed throughout the entire sample, whether in direct contact with the pressurizing medium or insulated from it in a flexible container. Thus, the process time is independent of sample size and shape, assuming uniform thermal distribution within the sample.
If a food product contains sufficient moisture, pressure will not damage the product at the macroscopic level as long as the pressure is applied uniformly in all directions. For example, a grape can be easily crushed if pressure is applied to it by placing it between two fingers and squeezing along one axis. In contrast, if the grape is exposed to a uniform pressure by submerging it in water inside a sealed flexible bottle and squeezing, the grape retains its shape no matter how hard the bottle is squeezed. In this case, the pressure transmitted from the bottle wall through the water is applied uniformly around the fruit. Similarly, pressure will not damage most foods processed by high pressure, providing the food does not have a porous structure containing air voids. Air pockets will collapse under pressure due to differences between the compressibility of air and water, and unless the food is perfectly elastic and consists of closed-cell foam from which air cannot escape, the food will not be restored to its original size and shape. As a result, foods like strawberries are crushed by HPP, but an air-filled balloon does not burst.
High-Pressure Equipment
Although the principles of high-pressure processing for microbial inactivation have been known since the late 1800s (Hite, 1899), it is only relatively recent developments in mechanical engineering that have permitted large high-pressure vessels to be constructed at reasonable cost with sufficient durability to withstand thousands of pressure cycles without failure. High-pressure processing systems were initially developed in the chemical and material process industries for applications such as making artificial diamonds and sintered materials from powders. It is only during the past two decades that the food industry has begun using pressure treatment for food preservation. HPP is primarily practiced as a batch process where pre-packaged food products are treated in a chamber surrounded by water or another pressure-transmitting fluid. Semi-continuous systems have been developed for pumpable foods where the product is compressed without a container and subsequently packaged “clean” or aseptically.
The primary components of an HPP system include a pressure vessel; closure(s) for sealing the vessel; a device for holding the closure(s) in place while the vessel is under pressure (e.g., yoke); high-pressure intensifier pump(s); a system for controlling and monitoring the pressure and (optionally) temperature; and a product-handling system for transferring product to and from the pressure vessel. Normally, perforated baskets are used to insert and remove pre-packaged food products from the pressure vessels. Systems also have provisions for filtering and reusing the compression fluid (usually water or a food-grade solution).
--- PAGE BREAK ---
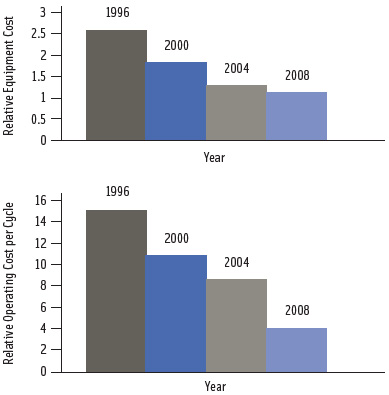
Commercial batch vessels have internal volumes ranging from 30 to more than 600 liters. Avure Technologies, NC Hyperbaric, and Uhde are major suppliers of commercial-scale pressure equipment. Both horizontal and vertical pressure vessel configurations are available. Commercial-scale, high-pressure processing systems cost approximately $500,000 to $2.5 million, depending on equipment capacity and extent of automation. Currently, HPP treatment costs are quoted as ranging from 4–10 cents/lb, including operating cost and depreciation, and are not “orders of magnitude” higher than thermal processing—as is often thought (Sàiz et al., 2008). As demand for HPP equipment grows, innovation is expected to further reduce capital and operating costs (Figure 1).
A typical HPP process uses food products packaged in a high-barrier, flexible pouch or a plastic container. The packages are loaded into the high-pressure chamber. The vessel is sealed and the vessel is filled with a pressure-transmitting fluid (normally water) and pressurized by the use of a high-pressure pump, which injects additional quantities of fluid. The packages of food, surrounded by the pressure-transmitting fluid, are subjected to the same pressure as exists in the vessel itself. After holding the product for the desired time at the target pressure, the vessel is decompressed by releasing the pressure-transmitting fluid (Farkas and Hoover, 2000). For most applications, products are held for 3–5 min at 600 MPa. Approximately 5–6 cycles/hr are possible, allowing time for compression, holding, decompression, loading, and unloading. Slightly higher cycle rates may be possible using fully automated loading and unloading systems. After pressure treatment, the processed product is removed from the vessel and stored/distributed in a conventional manner.
Liquid foods can be processed in a batch or semi-continuous mode. In the batch mode, the liquid product is pre-packaged and pressure-treated as described above for packaged foods. Semi-continuous operation requires two or more pressure vessels, each equipped with a free-floating piston that allows each vessel to be divided into two chambers. One chamber is used for the liquid food; the other for the pressure-transmitting fluid. The basic operation involves filling one chamber with the liquid food to be treated. The fill valve is closed and then pressure-transmitting fluid is pumped into the second chamber of the vessel on the opposite side of the floating piston. Pressurization of the fluid in this second chamber results in compression of the liquid food in the first. After an appropriate holding time, the pressure is released from the second chamber. The product discharge valve is opened to discharge the contents of the first chamber, and a low-pressure pump injects pressure-transmitting fluid into the second chamber, which pushes on the piston and expels the contents of the product chamber through the discharge valve. The treated liquid food is directed to a sterile tank from which sterile containers can be filled aseptically. Typically, three pressure vessels are used to create a semi-continuous system capable of delivering a continuous product output. This is accomplished by operating the three vessels such that one is loading, one is compressing, and one is discharging at any point in time (Farkas and Hoover, 2000).
The U.S. food industry has gained about 18 years of experience with commercial-scale, high-pressure equipment since the introduction of high-pressure pasteurization systems during the 1990s. In response to industrial demand for higher volume and throughput, high-pressure equipment vendors have focused on improving equipment productivity. Developments have included a doubling of pressure vessel volumes, automated systems for package and vessel loading, and higher horsepower intensifiers to speed up the compression process. Tandem machines (Sàiz et al., 2008), where multiple vessels are serviced by a common bank of high-pressure pumps, have also become available. By sequencing the compression cycles of multiple vessels, it is possible to increase throughput and reduce capital costs by shortening the compression time (through use of multiple pumps) and utilizing the same pumps to pressurize the other vessels during a period of time when the pumps would normally be idle in a single vessel system. With two or more pressure vessels operating under typical food processing conditions, a throughput of approximately 20 million lb/yr is achievable.
--- PAGE BREAK ---
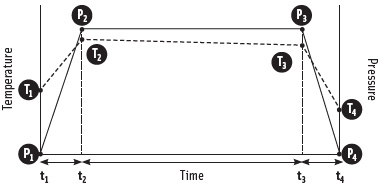
Pressure-Temperature Effects
During the compression phase (t1~t2) of pressure treatment, food products experience a decrease in volume as a function of the pressure (Figure 2). Both pure water and most moist foods subjected to a 600 MPa treatment at ambient temperature will experience about a 15% reduction in volume. The product is held under pressure for a certain time (t2~t3) before decompression (t3~t4). Upon decompression, the product will usually expand back to its initial volume (Farkas and Hoover, 2000). The compression and decompression can result in a transient temperature change in the product during treatment. The temperature of foods increases (T1~T2) as a result of physical compression (P1~P2). Product temperature (T2~T3) at process pressure (P2~P3) is independent of compression rate as long as heat exchange between the product and the surroundings is negligible. In a perfectly insulated (adiabatic) system, the product will return to its initial temperature upon decompression (P3-P4). In practice, however, the product will return to a temperature (T4) slightly lower than its initial temperature (T1) as a result of heat losses during the compression (elevated temperature) phase. The rapid heating and cooling resulting from HPP treatment offer a unique way to increase the temperature of the product only during the treatment, and to cool it rapidly thereafter.
The temperature increase of food materials under pressure is dependent on factors such as final pressure, product composition, and initial temperature. The temperature of water increases about 3°C for every 100 MPa pressure increase at room temperature (25°C). On the other hand, fats and oils have a heat of compression value of 8–9°C/100 MPa, and proteins and carbohydrates have intermediate heat of compression values (Rasanayagam et al., 2003; Patazca et al., 2007).
Microbial Efficacy
High-pressure treatments, in general, are effective in inactivating most vegetative pathogenic and spoilage microorganisms at pressures above 200 MPa at chilled or process temperatures less than 45°C, but the rate of inactivation is strongly influenced by the peak pressure (Patterson, 2005; Lau and Turek, 2007). Commercially, higher pressures are preferred as a means of accelerating the inactivation process, and current practice is to operate at 600 MPa, except for those products where protein denaturation needs to be avoided. The pressure resistance of vegetative microorganisms often reaches a maximum at ambient temperatures, so the initial temperature of the food prior to HPP can be reduced or elevated to improve inactivation at processing temperature (i.e., temperature at pressure). The extent of inactivation also depends on the type of microorganism, food composition, pH, and water activity. Gram-positive bacteria are more resistant than gram-negative, and generally speaking the larger and more complex the organism, the easier it is to inactivate. Significant variations in pressure resistance can be seen among strains of the same species (Cheftel, 1995). HPP causes damage to cell membranes and denatures some intracellular proteins leading to cell death. Many food components “protect” microorganisms from the effects of pressure treatment and it will often take a longer processing time or higher pressure to achieve the same degree of inactivation in food vs water or microbiological media (broths). Reduced pH is generally synergistic with pressure in eliminating microorganisms. Reduced water activity, however, tends to inhibit pressure inactivation with noticeable retardation as the water activity falls below ~ 0.95. Depending upon the pressure-temperature dose delivered, the process can also sub-lethally stress or injure bacteria. When validating an HPP pasteurization process, sufficient time after treatment should be allowed to confirm that sub-lethally injured organisms do not recover, and a nonselective microbiological media should be used to detect all viable organisms of concern during post-treatment storage.
Most yeast are inactivated by exposure to 300–400 MPa at 25°C within a few minutes, however, yeast ascospores may require treatment at higher pressures. Pressure inactivation of molds follows a pattern similar to yeast. Among viruses, the high degree of structural diversity is reflected in their wide range of pressure resistances (Smelt, 1998). While some human viruses appear more pressure-sensitive, other viruses such as polio virus are pressure-resistant. Most viruses can be eliminated by pressure treatments designed for eliminating bacteria of concern, but virus inactivation has not been as extensively researched as microorganisms.
--- PAGE BREAK ---
As mentioned earlier, bacterial spores can be difficult to inactivate using HPP, and require higher pressures, process temperatures, and holding times as compared to vegetative cells. Bacterial spores are often resistant to pressures above 1,000 MPa at ambient temperatures (Cheftel, 1995). Clostridium botulinum strains have been identified as the most pressure-temperature resistant pathogenic spores. Among the nonpathogenic spores studied, Bacillus amyloliquefaciens spores have been reported to have high pressure-heat resistance (Margosch et al., 2006; Ahnet al., 2007). More research is needed to characterize the combined pressure-thermal resistance of pathogenic and spoilage microorganisms as a function of the food matrix, pH, and water activity. Standardized reporting of process conditions, equipment, and microbial techniques utilized are needed in developing a comprehensive database of inactivation kinetics (Balasubramaniam et al., 2004). Additionally, molecular level mechanistic studies are essential for improving our understanding of combined pressure-thermal treatment effects on microorganisms. The extent and mechanisms of bacterial injury during high-pressure pasteurization and sterilization merit further investigation (Balasubramaniam and Farkas, 2008).
Commercial Applications
High-pressure processing provides a unique opportunity for food processors to develop a new generation of value-added food products having superior quality to those produced conventionally. Strawberry, apple, and kiwi jams represented the first wave of pressure-treated commercial products introduced into the Japanese market in 1990. Avocado-based products, especially guacamole, were subsequently commercialized in the United States. Fresherized Foods (formerly Avomex) began the first industrial production of guacamole in North America in 1997. A decade later, by 2007, approximately 120 industrial HPP installations were in use worldwide for commercial-scale food production (Sàiz et al., 2008). More than 80% of the equipment was installed after 2000, indicating an accelerated trend in HPP use. According to some industry estimates, high-pressure food pasteurization currently represents a $2 billion market (Hewson, 2008). North America (U.S., Canada, and Mexico), Europe (Spain, Italy, Portugal, France, UK, and Germany), Australia, and Asia (Japan, China, and S. Korea) lead the commercialization of high-pressure technology. The total production of pressure-treated food products is steadily growing. In 2008, it is estimated that 200,000 metric tons/yr (about 450 million lb/year) of pressure-treated products will be commercially available worldwide (Samson, 2008).
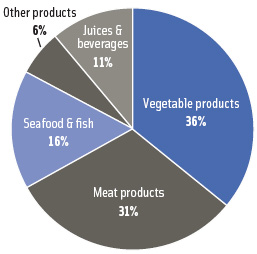
The novel nature of HPP technology and high equipment costs have been barriers to commercialization, but increased consumer demand for fresher-tasting foods containing fewer preservatives is driving increased usage. HPP can preserve food products without heat or chemical preservatives, and its ability to ensure safety and significantly extend refrigerated shelf life has opened new market opportunities particularly in the area of “natural” preservative-free meat products (Figure 3). Hormel Foods, Kraft Foods, Perdue, Foster Farms, and Wellshire Farms are examples of meat processors that have successfully utilized the technology for a variety of ready-to-eat, minimally processed meat products (Samson, 2008). Several seafood processors such as Motivatit Seafoods have also employed HPP to improve food safety and shelf life of shellfish with the added benefit of facilitating the removal of flesh from the shell. Other market segments employing pressure treatment include juice, beverage, and vegetable products.
The ability of HPP to deliver a “clean” ingredient statement, fresher flavor, and better nutrition provide a unique point of difference for producers. High-pressure processing is a paradigm-shifting technology for the food industry that is on-trend with consumer interests. Its use will likely grow as cost declines and food manufacturers identify new applications where HPP can deliver product quality improvements that consumers appreciate and will pay for.
V.M. (Bala) Balasubramaniam, a Professional Member of IFT, is Associate Professor of Food Safety Engineering, Dept. of Food Science and Technology, The Ohio State Univ., 2015 Fyffe Rd., Columbus, OH 43210-1007 (balasubramaniam.1@osu.edu), Daniel Farkas, a Professional Member of IFT, is Professor Emeritus, Dept. of Food Science and Technology, Oregon State Univ., Corvallis, OR 97331-6602 (dan.farkas@orst.edu), and Evan J. Turek, a Professional Member of IFT, is Senior Fellow with Kraft Foods, Kraft R&D Center, 801 Waukegan Rd., Glenview, IL 60025 (eturek@kraft.com). Send reprint requests to author Bala Balasubramaniam.
The high-pressure processing research laboratory (http://grad.fst.ohio-state.edu/hpp/) at The Ohio State Univ. was supported in part by the Ohio Agriculture Research and Development Center (OARDC), Center for Advanced Processing and Packaging Studies (CAPPS), and the food industry. References to commercial products or trade names are made with the understanding that no endorsement or discrimination by The Ohio State Univ. is implied.
References
Ahn, J., Balasubramaniam, V.M., and Yousef, A.E. 2007. Inactivation kinetics of selected aerobic and anaerobic bacterial spores by pressure-assisted thermal processing. Int. J. of Food Microbiol. 113(3): 321-329.
Balasubramaniam, V.M., and Farkas, D. 2008. High Pressure Processing. Food Sci. and Technol. Int. In press.
Balasubramaniam, V.M., Ting, E.Y., Stewart, C.M., and Robbins, J.A. 2004. Recommended laboratory practices for conducting high pressure microbial inactivation experiments. Innovative Food Sci. and Emerging Technol. 5(3): 299-306.
Balny, C. and Masson, P. 1993. Effects of high pressure on proteins. Food Rev. Int. 9(4): 611-628.
Black, E.P., Setlow, P., Hocking, A.D., Stewart, C.M., Kelly, A.L., and Hoover, D.G. 2007. Response of spores to high-pressure processing. Comp. Rev. Food Sci. Food Safety 6(4): 103-119.
Cheftel, J.C. 1995. Review: High pressure, microbial inactivation, and food preservation. Food Sci. and Technol. Int. 1: 75-90.
Dunne, C.P. 2005. High pressure keeps food fresher. Available at http://www.natick.army.mil/about/pao/05/05-22.htm. Accessed October 8, 2008.
Farkas, D. and Hoover, D. 2000. High pressure processing: Kinetics of microbial inactivation for alternative food processing technologies. J. Food Sci. (Supplement): 47-64.
Hayman, M., Baxter, I., Oriordan, P.J., and Stewart, C.M. 2004. Effects of high-pressure processing on the safety, quality, and shelf life of ready-to-eat meats. J. of Food Prot. 67(8): 1709-1718.
He, H., Adams, R.M., Farkas, D.F., and Morrissey, M.T. 2002. Use of High-pressure Processing for Oyster shucking and shelf-life extension. J. Food Sci. 67(2): 640-645.
Heremans, K. 2003. The effect of high pressure on biomaterials. In “Ultra High Pressure Treatments of Foods” ed. Marc E.G. Hendrickx and Dietrich Knorr. pp. 23-52. Kluwer Academic/ Plenum Publishers, New York.
Hewson, G. 2008. Personal communication. Avure Technologies, Kent, WA.
Hite, B.H. 1899. The effect of pressure in the preservation of milk. Washington, Va. Univ., Agriculture Experiment Station, Bulletin, 58: 15-35.
Hogan, E., Kelly, A., and Sun, D.W. 2005. High pressure processing of foods: An overview. In “Emerging technologies for food processing” ed. Sun, D.W., pp. 3-32. Elsevier Academic Press, London.
Lau, M.H. and Turek, E.J. 2007. Determination of quality difference in low-acid foods sterilized by high pressure versus retorting. In “High pressure processing of foods” ed. Doona, C.J. and Feeherry, F.E. pp. 195-217. Blackwell Publishing and IFT Press.
Margosch, D., Ehrmann, M.A., Buckow, R., Heinz, V., Vogel, R.F., and Gänzle, M.G. 2006. High-pressure-mediated survival of Clostridium botulinum and Bacillus amyloliquefaciens endospores at high temperature. Applied and Environ. Microbiol. 72(5): 3476-3481.
Matser A.M., Krebbers B., Berg, R.W., and Bartels, P.V. 2004. Advantages of high pressure sterilisation on quality of food products. Trends in Food Sci. and Technol. 15(2): 79-85.
Patazca, E., Koutchma, T., and Balasubramaniam, V.M. 2007. Quasi-adiabatic temperature increase during high pressure processing of selected foods. J. Food Eng. 80(1): 199-205.
Patterson, M.F. 2005. Microbiology of pressure-treated foods – A review. J. Applied Microbiol. 98(6): 1400-1409.
Rasanayagam, V., Balasubramaniam, V.M., Ting, E., Sizer, C.E., Bush, C., and Anderson, C. 2003. Compression heating of selected fatty food materials during high pressure processing. J. Food Sci. 68(1): 254-259.
Rastogi, N.K., Raghavarao, K.S.M.S., Balasubramaniam, V.M., Niranjan, K., and Knorr, D. 2007. Opportunities and challenges in high pressure processing of foods. Crit. Rev. in Food Sci. Nutr. 47(1): 69-112.
San Martin, M.F., Barbosa-Canovas, G.V., and Swanson, B.G. 2002. Food processing by high hydrostatic pressure. Crit. Rev. in Food Sci. Nutr. 42: 627-645.
Sáiz, A.H., Mingo, S.T., Balda F.P., and Samson C.T. 2008. Advances in design for successful commercial high pressure food processing. Food Australia, 60(4): 154-156.
Samson, C. Tonello. 2008. Personal communication. NC Hyperbaric, Spain.
Smelt, J.P.P.M. 1998. Recent advances in the microbiology of high pressure processing. Trends in Food Sci. Technol. 9(4): 152-158.
Tauscher, B. 1995. Pasteurization of food by hydrostatic high pressure: chemical aspects. Zeitschrift fuer Lebensmittel Untersuchung und Forschung 200: 3-13.
Torres, J.A. and Velazquez, G. 2005. Commercial opportunities and research challenges in the high pressure processing of foods. J. Food Eng. 67(1-2): 95-112.
Authors
-
V.M. (BALA) BALASUBRAMANIAM, DANIEL FARKAS, , EVAN TUREK
Categories
-
Food Processing and Packaging
-
Food Sciences
-
Food Safety and Defense
-
Nonthermal Processing
-
Food Microbiology
-
Food Technology Magazine



