Is the Future of Meat Animal-Free?

What is clean meat? And why is this field emerging now? Clean meat (also called cultured meat) is the term for genuine animal meat grown by farming cells directly rather than via rearing and slaughtering an animal. At its essence, meat is simply a collection of cells—predominantly muscle cells, with fat cells and a handful of other cell types, collectively called the connective tissue, contributing to its structure. All of these cells can be grown in controlled, sterile environments outside of the body of an animal, resulting in a product that replicates the sensory and nutritional profile of meat.
This idea has been contemplated for nearly a century, but only in recent years have tools been developed that enable the earnest pursuit of clean meat at commercial scale. Many of the underlying technologies stem from unrelated fields that utilize animal cell culture, most notably, tissue engineering, biopharma, and cell-based therapeutics.
Cell culture advances from the biomedical realm are now being applied to clean meat research, just as industrial biotechnology advances have been translated from biomedical applications (for example, using yeast hosts to produce low-cost insulin) into large-scale applications for food (utilizing essentially the same production platform for food processing aids like “vegetarian rennet” and other enzymes). This long history of translational R&D effectively provides the clean meat field with an accelerated path to commercialization because it leverages decades of research and billions of dollars of public investment into relevant fields like stem cell biology and developmental biology. This work has enabled a detailed understanding of the basic biology underlying the growth and differentiation of cells outside of the context of their native animal systems.
A Broad Array of Benefits
What are the benefits of clean meat relative to industrial animal meat production? The benefits to society and the environment conferred by transitioning from industrial animal meat production to clean meat production at a global level are likely the most compelling even if consumer considerations will ultimately be the largest drivers of future demand for these products. The global-scale advantages of clean meat can be categorized as greater food security and global resource equity, significant environmental and sustainability benefits, and decreased public health risks.
Because clean meat is grown in a closed, controlled environment, it can be produced without antibiotics and risk of contamination with bacteria like Salmonella and E. coli, common culprits of serious foodborne illness. Correspondingly, preliminary data show that the shelf life of clean meat is significantly longer than that of industrially produced meat because no bacteria are present to accelerate the degradation process. This translates into less food waste at every step of the supply chain—from manufacturers to grocers to consumers. Additionally, clean meat does not contribute to the current distortions in global grain markets that result from the massive feed requirements of livestock animals, which are fed predominantly to consumers in wealthy countries. Thus, a more efficient production method promotes food security at home and abroad.
From an environmental perspective, the current life-cycle analyses of clean meat rely on preliminary assumptions, but estimate order-of-magnitude or greater reductions in water footprint, land use, and greenhouse gas emissions relative to industrial meat production. The increased efficiency—and thus lower resource requirements across the board—of clean meat is due to fundamental thermodynamics: this production method does not rely on shuttling calories through the metabolizing, moving, breathing, heat-generating body of an animal in order to obtain a small fraction of those inputs as meat. At scale, production of clean meat is predicted to be inherently more sustainable than current industrial animal agriculture.
Finally, clean meat addresses two public health threats associated with animal agriculture that are routinely downplayed relative to their enormous potential impact: bacterial antibiotic resistance and outbreaks of zoonotic disease (infectious diseases that jump from animals to humans). Both of these phenomena have a high rate of emergence from concentrated animal farming operations, and specific antibiotic resistance genes and individual strains of zoonotic diseases can often be traced directly to animal farms. In an increasingly globalized world with a dwindling number of effective antibiotics in our arsenal, these threats pose a very real risk of catastrophic consequences. This prompted the World Health Organization (WHO) to issue recommendations in November 2017 to reduce antibiotic use in farmed animals, with Kazuaki Miyagishima, WHO’s director of the Dept. of Food Safety and Zoonoses, stating, “Scientific evidence demonstrates that overuse of antibiotics in animals can contribute to the emergence of antibiotic resistance. The volume of antibiotics used in animals is continuing to increase worldwide, driven by a growing demand for foods of animal origin” (WHO 2017). Clean meat can eliminate both animals and antibiotics from the food production process altogether.
Producing Clean Meat
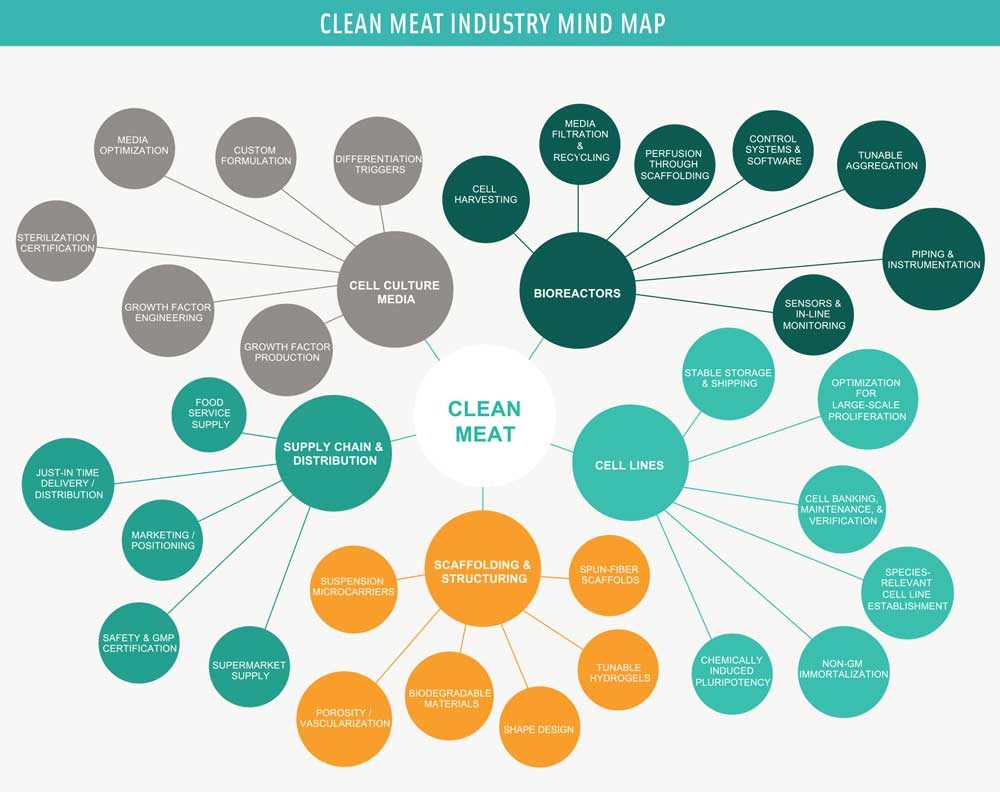
What will large-scale production of clean meat look like? The technological challenges for clean meat can be grouped into four critical technology areas: cell line development, cell culture nutrient media, scaffolding and product structuring, and the bioreactors in which the process is housed. While no production-scale or pilot-scale facilities yet exist for clean meat, significant advances in all of these areas toward large-scale cell culture for cell therapies and tissue engineering give us insight into what the clean meat production process may look like at scale. The process can be conceptualized as two phases: the first is the proliferation phase, in which cells divide and multiply to increase in mass, and the second is the differentiation or maturation phase, during which cells transition from proliferative cells into the desired final cell types (muscle fibers, fat cells, etc.). Each of these phases involves slightly different design requirements for each of the four critical technology elements.
• Cell Line Development. Cell lines form the starting culture from which large quantities of cells multiply. Initially, donor cells are harvested from an animal either through live biopsy, immediately after slaughter, or from an embryo. After the initial harvest, there are a number of methods for sustaining the cells’ capacity to proliferate continuously, thereby reducing and ultimately eliminating the need to isolate new cells. Companies are exploring several types of cells in various stages of differentiation to determine which cells exhibit optimal performance for both proliferation and maturation into the desired final cell types.
Some groups are working with stem cells that have inherently high proliferative capacity and that are capable of differentiating into multiple cell types that are relevant for meat (for example, muscle, fat, and connective tissue). Others are working with so-called adult stem cells that have less flexibility but that differentiate into the desired cell type with higher efficiency. It remains to be seen whether one approach will win out at scale, or whether these diverse strategies will continue to be used by various companies.
While some companies are exploring genetic modification and genome editing techniques to enhance the performance of their cell lines, genetic engineering is by no means necessary for large-scale animal cell culture; it is simply one tool in the toolkit for optimizing the efficiency and robustness of the process.
• Cell Culture Media. Cells require nutrient media, a liquid broth containing salts, sugars, and amino acids as well as signaling molecules called growth factors, for their proliferation and their maturation. Currently, this media is rather expensive because the existing clients of such media (biomedical research and clinical cell therapies) require pharmaceutical-grade component sourcing and even the largest-scale applications are purchasing small volumes relative to the scale that would be required for meat production. Scaling projections indicate that the cell culture media could rather easily be made orders of magnitude less expensive than the current market price. This is significant for the commercial feasibility of clean meat as a whole, as media is its primary input and the cost of the media is anticipated to be the main cost driver of the final product.
• Scaffolding and Product Structuring. In order to form a tissue-structured piece of meat, the cells are seeded onto a scaffold material that provides a physical support structure as they differentiate and mature into the desired cell types (muscle, fat, etc.). At its simplest, the scaffold is simply an inert material to which the cells adhere, but more sophisticated scaffolds may help guide the cells into forming the final product.
There are several options for scaffolding materials for clean meat products that require a scaffold (as noted below, some products may not require scaffolding at all if the structure will be imparted through downstream processing). Scaffolds may be made of edible, food-grade, taste-neutral materials that will still be present at some fraction in the final product. Alternatively, they may be made of biodegradable materials that the cells will break down as they grow and which then will be replaced by the cells’ own extracellular matrix (a network of proteins and support structures).
In either case, the scaffolding material must be sufficiently porous to allow the nutrient media to flow through it and feed all of the cells. It is also possible to use the biomechanical properties of the scaffold (parameters like stiffness and curvature) to guide the differentiation of the cells, giving some degree of spatial control over where cells differentiate into various types: picture a marbled steak with heterogeneous patterns of fat and muscle cells.
• Bioreactor Design. The bioreactors house this entire process in closed containment from start (inoculation of the seed culture) to finish (harvesting an intact piece of meat). There may be several different types of bioreactors needed depending on the phase (proliferation versus maturation) and the desired final product type (a minced meat product versus a thick cut). Most companies aim to incorporate media monitoring so that additional nutrients can be added as they are depleted by the cells while retaining and recycling the other components in order to reduce costs. In addition, as the process scales, everything from media addition to product harvesting will have to be automated to improve process robustness and decrease labor costs.
The Clean Meat Landscape
The current competitive landscape consists of nearly a dozen start-up companies, almost all of which have formed within the past 18 months. These companies span the globe, illustrating the international scope of this endeavor. The list of companies pursuing this technology includes three in Israel, at least one in Europe, at least three in the United States, and a few emerging in Australasia and East Asia. This landscape is likely to become significantly more populated in the coming year as companies take shape, focusing on new product categories, new process innovations, or new technological solutions to challenges facing the entire field.
In the United States, Memphis Meats is the furthest along in terms of demonstrated product yield and investment raised to date. In August of 2017, the company closed its Series A funding round with lead investor DFJ—which is perhaps best known for its leading-edge futuristic technology investments in companies like Tesla and SpaceX—and additional investment from meat conglomerate Cargill. This represents the first publicly disclosed investment of a conventional meat producer in the clean meat field, but other start-ups are engaging in discussions with meat companies as investors or partners at even earlier stages.
It could be argued that existing meat industry partners have an incentive to keep a close eye on this emerging field from a competitive threat perspective. However, proactive members of the industry are recognizing the potential enormous benefits clean meat confers to producers, including more stable supply and shorter production cycles (leading to less price volatility and not subject to the whims of infectious disease outbreaks that have the potential to spread quickly through industrial animal farms).
From a meat science perspective, the degree of precision and control offered by the clean meat production process means that only meat of the highest quality would be produced. The attempt to valorize low-quality cuts or byproducts would become a thing of the past, as these side-streams are also prone to disruption from other cellular agriculture or plant-based innovations (for example, cellular agriculture company Geltor producing high-quality, end-use-tailored gelatin).
A shift toward clean meat also represents an opportunity to mitigate the significant risks the meat industry currently faces as a result of external factors and the heavy resource burden of goods. A coalition of global investors representing more than $2.3 trillion of assets under management has outlined these risks, led by the FAIRR (Farmed Animal Investment Risk and Return) Initiative. Given the shifting political tides in some jurisdictions toward government incentives for sustainable business practices and harsh penalties for externalized environmental costs, the inherent inefficiencies of animal agriculture create an unstable business model that is subject to market disruption caused by policy departures from the status quo.
Bringing Clean Meat to Market
When will these products appear on the market? When assessing which clean meat products may come to market first and when we should expect to see them on supermarket shelves, it is helpful to consider two critical aspects: one is the price the consumer will pay and the other is the level of sophistication of the product. Many of the existing start-up companies anticipate that their first product will arrive on the market somewhere in the time frame of three to five years, and that it will be introduced at a premium price point—perhaps even through high-end restaurants. This is a go-to-market strategy not unlike that of Impossible Foods, which debuted its heme-enriched plant-based burger in a limited number of restaurants in 2016 prior to a larger market rollout.
With regard to the level of technological sophistication, products that mimic processed meats or meats with relatively homogenous, non-complex structures will be easier to produce. A product like a sausage, a burger, a fish stick, or a chicken nugget will be much easier to mimic than a product like a marbled steak or an intact chicken breast. These processed products exhibit a structural complexity that is readily achieved from small pieces of cultured tissue, rather than requiring a complex, intact tissue containing multiple types of co-cultured cells. Furthermore, as with the industrially produced animal versions of these processed meats, these products contain a significant fraction of non-meat binder or filler ingredients that can improve sensory aspects of the product.
While some companies are pursuing specific types of meat at the species level (Bay Area start-up Finless Foods has focused exclusively on finned fish, for example), many companies are developing malleable platforms that can be adapted to accommodate cells derived from various species. The biological needs of specific cell types, such as muscle cells, are strikingly similar regardless of the species of origin across the animal kingdom. Accordingly, a company that excels in developing a production platform for a ground beef product will likely be able to produce a ground chicken or ground turkey product with relatively little adaptation. Likewise, a company specializing in more complex, tissue-structured products that involve simultaneous co-culture of fat cells and muscle cells in a designated pattern is likely to excel at both marbled steaks and cuts of fatty pork. A company whose platform is ideal for cultivating thin slices of tissue may delve into both the bacon and the sashimi market in quick succession.

A Look at Challenges and Opportunities
Several companies have demonstrated public tastings of their meat: Mosa Meat’s founders debuted the first burger in 2013; Memphis Meats has showcased everything from a meatball to fried chicken to duck a l’orange; Finless Foods cooked up clean fish at its IndioBio demo day; and the team behind Toyko-based Integriculture has sampled miniature foie gras. Clean meat has been proven in principle, and the predominant challenges revolve around scale and cost. However, fundamental research questions remain around maximizing the growth efficiency of the cells, modulating their differentiation into muscle and fat, developing scaffolds that facilitate thick cuts of meat, and growing multiple cell types simultaneously.
These projects are being tackled through a combination of academic research, research underway at clean meat start-ups, and emerging partnerships with existing food and life science industry players that are keen to contribute their expertise. As the competitive landscape becomes increasingly populated, it is anticipated that this development will continue to accelerate as the field becomes an attractive opportunity for new companies to provide supporting services or raw materials.
The IFTNEXT initiative is a heightened, purpose-drive commitment to bringing provocative ideas and discoveries together to inspire thoughtful, important conversations that challenge conventional approaches with the goal of informing global issues related to the science of food.
Liz Specht, PhD, a member of IFT, is a senior scientist with The Good Food Institute, a nonprofit organization that supports the growth of plant-based and clean meat alternatives to industrial animal agriculture (lizs@gfi.org).
IFTNEXT is made possible through the generous support of Ingredion, IFT’s Platinum Innovation Sponsor.
Hero Image: From Adaptogens: Herbs for Longevity and Everyday Wellness 2019. @ Homunkulus28/iStock/Getty Images Plus, @ Awispa/iStock/Getty Images Plus, @ Marakit_Atinat/iStock/Getty Images Plus, @ Veleri/iStock/Getty Images Plus, @ cuttlefish84/iStock/Getty Images Plus
Authors
-
Liz Specht
Categories
-
Emerging Science and Technologies
-
Food Product Development
-
Food Ingredients and Additives
-
Cultured Meat
-
Proteins
-
Food Technology Magazine



