Enzymes: Tools for Creating Healthier and Safer Foods
Advances in enzyme technology help food technologists create foods that address the global concerns of obesity, malnutrition, and food safety.
The food industry is facing some major challenges. Malnutrition, both over- and under-nutrition, continues to affect populations in developed and developing countries, resulting in increased disease and often catastrophic loss of life.
 The increased consumption of energy-dense foods with high levels of sugar and saturated fats, combined with reduced physical activity, has led to an increase in obesity. The World Health Organization estimates that 300 million people in the world are clinically obese (WHO, 2004). In the United States, obesity could soon overtake tobacco as the leading cause of preventable death (Mokdad et al., 2004).
The increased consumption of energy-dense foods with high levels of sugar and saturated fats, combined with reduced physical activity, has led to an increase in obesity. The World Health Organization estimates that 300 million people in the world are clinically obese (WHO, 2004). In the United States, obesity could soon overtake tobacco as the leading cause of preventable death (Mokdad et al., 2004).
Fueled by the success of the Atkins and other related low-carb diets, millions of Americans are now restricting their intake of carbohydrates. As a result, the food industry is rapidly reformulating its products to better address the needs of consumers desiring foods and beverages with fewer carbohydrates. Although low-carb foods may not be the ultimate answer to weight management, it is possible that this sudden change in consumer behavior could be symptomatic of a far greater unmet need for foods that not only taste good but also positively contribute to oneb�19;s health and well-being. These are major challenges for the food industry, but more important, they offer a great opportunity for the industry to develop solutions to these problems in the form of healthier and safer foods and beverages.
Enzymes are the perfect tools for food formulators to address some of these problems. They are specific in their action on substrates relative to chemical and physical processing of foods and beverages. They safely and effectively enable food technologists to selectively modify carbohydrates, proteins, and lipids in complex food systems to create foods that taste good, improve health, and are safe to consume.
For example, if one wants to selectively modify a specific protein within a complex food system containing other proteins, carbohydrates, and lipids, an enzyme that breaks a specific amino acid bond may prove to be more effective than a process such as heating or the addition of a chemical oxidant that might affect all the food substrates in various and often uncontrolled ways. In addition, enzymes, being biological proteins with a safe history of use, are usually regarded as label-friendly processing aids in foods and beverages, in comparison to many chemical additives.
Today, as food and beverage processors design new products that will satisfy the growing consumer need for healthy and safe foods and beverages, enzymes are often proving to be the preferred means with which food processors can achieve their objectives. This article explores the potential use of enzyme technology to process foods and assist formulators in creating healthier and safer foods that address the global concerns of obesity, malnutrition and food safety.
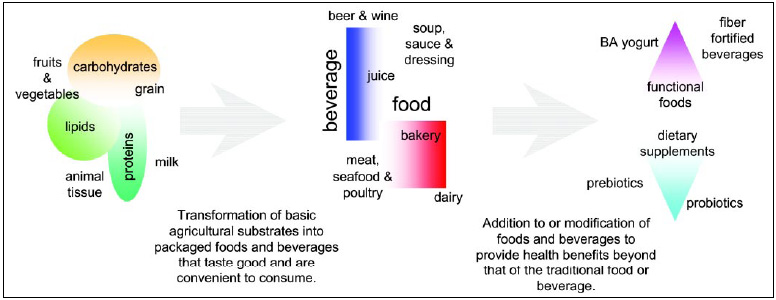
Modifying Carbohydrates
Modern enzyme technology involving the use of exogenous enzymes was initially adopted for the hydrolysis of starch. Exogenous enzymes are tightly integrated into processes for starch hydrolysis and associated products (maltodextrins, glucose syrups, very-high-maltose syrups and high-fructose syrups). The routes to produce these materials and the enzymes employed are well established (Bentley and Williams, 1996). Much of the progress in using enzymes in food processes or in the manufacture of food ingredients has been evolutionary, involving improvements in catalytic activities, the ability to deal with a wider range of raw materials, wider variations in processing conditions, pH, and temperatures, and reduction in the cost of enzymes.
--- PAGE BREAK ---
Many of these improvements have been brought about by the introduction and use of enzymes from genetically modified microorganisms (GMOs). In addition, there have been new developments that involve new enzyme activities and new applications for enzyme activities. Many of the recent and on-going developments for use of enzymes in starch hydrolysis are associated with improvements in enzyme activities (�1;-amylases, glucoamylases, and debranching enzymes) and improvements in enzymatic hydrolysis of non-corn cereals such as wheat and barley.
Employing enzymes has also made the use of traditional products for new purposes possible. For example, maltose syrups, traditionally produced for brewing applications, are now used for the production of maltitol, which can replace sugar one-for-one, with a relative sweetness of 90%. In most traditional recipes, such as cereals, milk products, dry mixes, tablets, snacks, and marzipan, maltitol can now be substituted for sucrose. The introduction of the enzyme inulinase has allowed the use of fructan root crops (chicory and the Jerusalem artichoke) for the production of fructans and fructose (Fleming and Groot Wassink, 1979; Fuchs, 1993), which can serve as dietary fibers and low-calorie sweeteners.
The use of cyclodextrins (Hedges, 1998) as food additives with applications as carriers and stabilizers of flavors, colors, and some vitamins is possible as a result of cyclodextrin glycosyltransferase, a starch-degrading enzyme that converts starch into a mixture of cyclic malto-oligosaccharides (cyclodextrins). Because they have a hydrophilic outside and a hydrophobic cavity that enables them to form specific inclusion complexes with small hydrophobic molecules, cyclodextrin complexes may have increased thermal and oxidative stability and may be used to reduce irritative side effects and mask tastes and odors.
Within the brewing industry, increases in demand for process efficiency, solutions to process difficulties, consistency of a particular product, and brewing with adjuncts (the inclusion of carbohydrate sources other than malted barley) have resulted in the use of exogenous enzymes. Consequently, �1;-amylase,-amylase, glucoamylase, -glucanase (endo-1,3(4)--glucanases), and proteinase activities are used in brewing (Ob�19;Rourke, 1996).
Developments in the brewing process and the requirement for the product to meet changing consumer expectations are being met by new uses of enzymes. Most recently, interest has been shown in developing beers in which the wort fermentability has been adjusted or controlled. The production of reduced-fermentable beers that contain low or no alcohol is being achieved by using a combination of highly specific thermo-stable enzymes (e.g., Genencorb�19;s AmylexB. 3T and Beta Glucanase 750L) that can ensure adequate starch release and break-down and, at the same time, reduce wort viscosity, resulting in a higher than normal level of unfermentable oligosaccharides.
On the other hand, production of super-attenuated lite beers containing very low carbohydrate content (�3; 2.6 g of carbohydrate/275 mL) but still having 5% abv (alcohol by volume) can be achieved with the use of enzymes such as glucoamylase (Matthews et al., 2001) and other debranching enzymes (e.g., pullulanase). Conventional worts contain nonfermentable dextrins, as malt enzymes cannot hydrolyze all the �1;(1b�6;�12;6) bonds in the amylopectin during mashing. Glucoamylase increases the fermentability of the wort by hydrolyzing the �1;(1b�6;�12;4) and, more slowly, �1;(1b�6;�12;6) bonds in the dextrins, and pullulanase specifically hydrolyzes the �1;(1b�6;�12;6) bonds. Wort fermentability can also be increased by the addition of glucose syrups (Strauss et al., 1979).
--- PAGE BREAK ---
The benefits of producing beer with substantial levels of nondigestible soluble dietary fiber that can be used by the microflora in the lower intestine have also been recognized. Soluble dietary fiber in the form of isomalto-oligosaccharides (and fructo-oligosaccharides), having a degree of polymerization of 4 or more, can be produced in the brewing process by the action of D-glucosyltransferase. High-gravity worts that have a higher ratio of maltose and maltotriose to glucose favor the formation of isomalto-oligosaccharides. The same effect can also be achieved by the direct addition of isomalto-oligosaccharide syrup to the fermenter (Brier et al., 2002).
An important objective in the juice industry is to process materials at a low cost while maintaining or improving the organoleptic properties and stability of the finished product. Exogenous enzymes allow efficient extraction of components and are used to degrade the cell wall of fruits for pulp maceration, liquefaction, and juice depectinization. A wide range of pectinolytic activities, hemicellulases, and cellulases involved in commercial applications have been reviewed by Grassin and Fauquembergue (1996). In addition, acid �1;-amylases are used for hydrolysis of fruits containing starch.
One of the benefits of fruit and vegetable consumption is the ingestion of flavonoid glycosides (glycosides of polyphenols). A high intake of these has been linked to a variety of health benefits, including beneficial action on gut microflora, antimicrobial action (e.g., inhibition of Listeria monocytogenes), a lower incidence of colon cancer (and possibly other cancers), and decreased cardiovascular disease. During enzymatic treatment of fruits and vegetables, one of the challenges for the food technologist is to optimize the bioavailability of these materials in their most effective form.
The trend for low-carbohydrate diets has been noted in the production of super-attenuated beers. Another trend is in foods that promote a healthy digestive system. Traditionally associated with high-fiber foods, a healthy digestive system is also being linked to the consumption of probiotics and prebiotics. Prebiotics are defined as nondigestible food substances that stimulate the growth and activity of beneficial bacteria in the lower intestine. Nondigestible oligosaccharides and resistant starch are examples of prebiotics.
Modifying Proteins
Traditionally, proteolytic enzymes have been used to modify food proteins to improve their flavor, texture, functionality, and nutritional quality. Examples include the use of exopeptidases to reduce bitterness in enzyme-modified cheese and bacterial proteases to restore emulsification properties to heat-denatured soy proteins. More recently, proteolytic enzymes are being used to improve the health and safety aspects of food proteins. Proteases are providing food technologists tools to improve the palatability of reformulated low-carb/high-protein foods. Enzyme-modified proteins replace fats and carbohydrates by providing texture, viscosity, mouthfeel, flavor (especially sweetness), and brothy notes. Proteins with altered fat-and water-binding properties have enhanced the ability to replace fats and carbohydrates in food products. Moreover, they can contribute to the healthfulness of foods. Two classic examples are SimplesseB., a microparticulated whey protein product from CP Kelco, and Dairy-LoB., a thermally denatured whey protein product from Cultor Food Science. Both these products have achieved a level of market acceptance as fat replacers in dairy foods, salad dressings, sauces, and other foods.
Proteases are also used to produce bioactive peptides with various functionalities, reduce allergenicity of food proteins, and protect food quality. The human body produces a wide array of bioactive peptides which function as biological messengers, stimulating or suppressing a wide range of physiological responses. Evidence now suggests that specific peptides derived from food proteins also act as messengers and play a significant role in maintaining health and preventing disease (Gauthier and Pouliot, 2003). These bioactive peptides can be produced in vitro by enzymatic hydrolysis of food proteins such as casein, eggs, wheat gluten, soy, and whey proteins.
--- PAGE BREAK ---
At least 40 different food-derived peptides have been shown to produce an anti-hypertensive effect. These peptides, ranging from 2 to 30 amino acids, inhibit angiotensin-converting enzyme (ACE) and can be produced by enzymatic hydrolysis of whey proteins and casein. An example of a commercial product containing whey-derived ACE-inhibitory peptides is BioZateB. 1 produced by Davisco Foods International. Calpis in Japan and Evolus in Finland, among others, also produce products with ACE-inhibitory claims.
Food-derived bioactive peptides have also been shown to contribute to weight management. A specific casein peptide, glycomacropeptide (GMP), plays a significant role in appetite suppression and is produced by the hydrolytic action of chymosin or other proteases on �4;-casein. It stimulates the production of cholecystokinin, an intestinal hormone, which induces the sensation of satiety (Brody, 2000). Arla Foods and Nutricepts are companies that promote GMP-enriched whey products. Similar beneficial weight management effects have been demonstrated with peptides derived from proteolytic hydrolysis of soy protein isolates.
Many protein-rich foods such as fish, seafood, peanuts, tree nuts, soy, wheat, and milk proteins can trigger allergic reactions in susceptible individuals. Specific amino acid sequences (epitopes) present in the food proteins interact with cells of the immune system to trigger an allergic response. Hydrolysis with proteolytic enzymes, physical removal, chemical modification, and heat denaturation are some of the potential strategies used for elimination of allergenic epitopes.
Treatment of chickpea protein isolates with a combination of endo- and exopeptidases has been shown to result in a significant reduction in allergenicity without any effect on nutritional quality (Clemente et al., 1999). In addition, a combination of thermal treatment and proteolytic hydrolysis was shown to significantly decrease the allergenicity of bovine -lactoglobulin (Iametti et al., 2002).
On the other hand, wheat protein epitopes associated with celiac disease, a relatively common food allergy, are highly resistant to proteolytic hydrolysis. At this time, the only reliable method to prevent symptoms is complete avoidance of gluten-containing food products. However, there is hope that the application of proteolytic enzymes (in vitro or in vivo) may help eliminate or reduce the symptoms associated with celiac disease (Anderson et al., 2000).
Enzymes and various antimicrobial peptides have also been effectively used to reduce pathogens in foods and to extend the shelf life of many perishable foods. The antimicrobial peptide nisin has been used to prevent the growth of botulinum spores in processed cheese and, more recently, in combination with the enzyme lysozyme, has demonstrated efficacy in killing L.monocytogenes on the surface of cooked meat products such as hot dogs (King et al., 2003).
While chemical antioxidants have a long history of use in food systems, they are perceived as chemical additives by consumers, and their use has raised health concerns. Enzymes offer an alternative approach to food protection. Glucose oxidase and catalase function as a very powerful oxygen-scavenging system in packaged foods and beverages. When sufficient glucose levels are present, glucose oxidase and catalase remove 100% of the available oxygen from an enclosed system, thereby preventing the flavor and color defects associated with oxidation and spoilage resulting from mold growth (Min et al., 2003). Glucose oxidase can also be used as a glucose scavenger in food products. For example, it is used to desugar egg whites prior to drying, preventing the undesirable Maillard browning during the drying process.
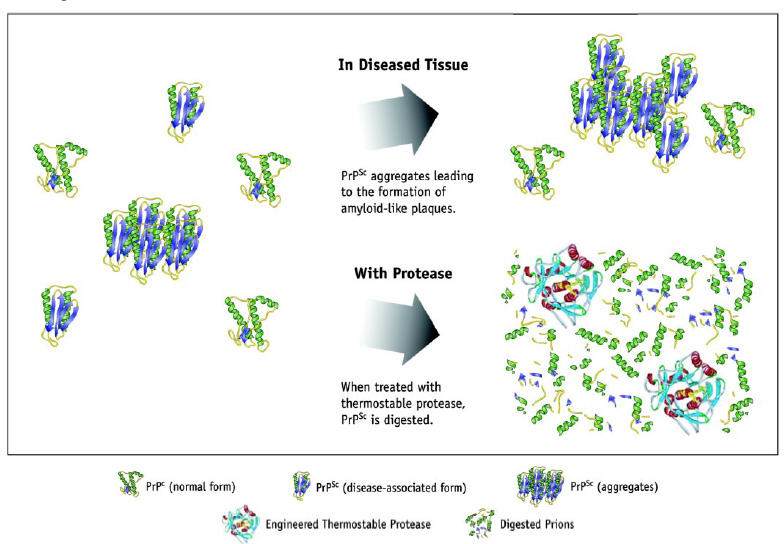
Recently, enzymes have also found an application in reducing the infectivity of prions associated with bovine spongiform encephalopathy (BSE) and other related disorders such as variant Creutzfelt-Jacob disease (vCJD). These relatively new additions to the list of foodborne illnesses are caused by a particle known as a prion (proteinaceous infectious particle), an incorrectly folded version of a neural cellular protein. There is no known cure for BSE, and the BSE prion has proven resistant to chemical and thermal treatment and hydrolysis by most proteolytic enzymes.
In collaboration with the United Kingdomb�19;s Health Protection Agency, Genencor International has demonstrated the efficacy of a proprietary thermostable, protein-engineered endopeptidase for significantly decreasing the infectivity of the BSE prion in contaminated bone meal (Genencor, 2004). In addition to medical applications, this work has numerous potential applications in the meat processing industry.
--- PAGE BREAK ---
Modifying Lipids
Lipids and phospholipids are found in every living tissue, particularly in cell membranes. Thus, it is not surprising that these fats are a critical part of human nutrition and health and a factor in the formulation of many foods. Enzyme technology is helping to increase the functionality and healthfulness of these basic raw materials.
Growing evidence has suggested that dietary choline and lecithin positively affect human health in various ways. Lecithin, a fatty substance manufactured in the body and widely found in many animal- and plant-based foods, including eggs, liver, peanuts, soybeans, and wheat germ, is considered an excellent source of choline. Phospholipases (such as PLA 1, 2, C, and D), enzymes that normally breakdown ingested lipids in our digestive tracts, are currently used as aids in food processing to extract phospholipids from lecithin-rich foods. In 1996, the Food and Drug Administration affirmed that lysolecithin prepared by the action of PLA2 on lecithin was generally regarded as safe (GRAS) for use as a direct food ingredient (FDA, 1996).
In addition to the health-related effects, the food industry has long recognized the importance of lecithin as a critical component in food processing. Lecithin helps smooth the texture of food items such as chocolate and margarine and makes instant foods, such as infant formulas, easy to dissolve.
Enzymes are also being used to modify fats to reduce their calorie content. Kao Corp. has developed a healthier fat composition for converting the triglycerides in common oil into diacylglycerides (DAGs), employing enzyme technology (Masui et al., 2003; Koike et al., 2003). DAGs have been shown to reduce arteriosclerotic factors in blood, as well as inhibit various degenerative diseases, an effect thought to occur because DAGs are metabolized differently than triglycerides, causing them to be burned quickly as energy instead of contributing to the deposition of adipose tissue. However, DAGs perform the same as conventional fats in cooking and frying, baking, salad dressings, and dairy-based products.
The DAG production process relies on an enzyme called 1,3-specific lipase to catalyze the customized addition of fatty acids to a glycerol backbone. The enzyme may be immobilized using technologies known to the enzyme industry, offering the advantage of repeated enzyme use (Scheels, 2002).
The flight away from trans fats is fueling interest in alternatives, and once again enzymes are playing a role in their development. Catalytic hydrogenation of unsaturated vegetable oils increases their degree of saturation, giving them properties resembling animal fats. The chemical catalyst typically used is not specific; and if the hydrogenation is not complete, this will result in formation of unnatural trans double bonds in addition to natural cis-configured bonds. The amazing specificity of enzymes can replace this random catalysis with the specific formation of only cis bonds, thereby reducing the formation of the trans bonds.
--- PAGE BREAK ---
Another enzymatic approach, one more likely to enjoy commercial acceptance, is the use of lipases to catalyze interesterification. This catalytic method of redistributing the fatty acids on the glycerol portion is another way to increase functionality of oils without negatively affecting healthfulness. Unsaturated fatty acids attached to a triglyceride in a vegetable oil could be replaced with fully hydrogenated, saturated fatty acids prepared by conventional hydrogenation. This would allow building a custom-made triglyceride with the desired degree of saturation but no trans fat. These processes could also benefit from using immobilized lipases.
Just the Beginning
Enzymes have been used safely in a wide variety of foods for centuries. The biodiversity of enzymes is providing the food industry with a wide range of functionalities. As biotechnology paves the way for making improvements to known enzyme functions as well as opening the door for designing new enzymes with added functionalities, the GMO issue may continue to be one of controversy for the foreseeable future. The food industry has already benefited from several enzymes that are produced using genetically modified production hosts to reduce the cost or enhance the functionality of the enzyme. Future biotechnology applications will more likely focus on engineering enzymes to provide benefits that are valued by consumers, such as those that directly affect the health and safety of the consuming public.
Used in this fashion, it is more likely that consumer acceptance of GMO solutions will increase as the perceived risks fall below the benefits to be gained by consumers. Then, enzymes improved with biotechnology will be more widely used by food and beverage processors to create products that help address some of the problems affecting our world today.
by Peter Birschbach, Neville Fish, Wayne Henderson, and Douglas Willrett
The authors are, respectively, Senior Manager, Technical Services; Director, Global Applications and Technical Services; Manager, Market Development; and Director, Food and Beverage, Genencor International, Inc., 200 Meridian Centre Blvd., Rochester, NY 14618. Send reprint requests to author Willrett ([email protected]).
References
Anderson, R.P., Degano, P., Godkin, A.J., Jewell, D.P., and Hil, A.V. 2000. In vivo antigen challenge in celiac disease identifies a single transglutaminase-modified peptide as the dominant A-gliadin T-cell epitope,b�0;0;0;0;0;0;0;0;0;0;0;1D; Nat. Med. 6: 337-342.
Bentley, I.S. and Williams, E.C. 1996. Starch conversion. In Godfrey and West (1996), pp. 339-357.
Brier, M., Shetty, J.K., and King, J.W. 2002. High soluble dietary fiber fermented beverage and process for its production. PCT #WO 0220712 A 1.
Brody, E.P. 2000. Biological activities of bovine glycomacropeptide. Brit. J. Nutr. 84(Suppl.): S39-S46.
Clemente, A., Vioque, J., Sanchez-Vioque, R., Pedroche, J., and Millan, F. 1999. Effect of enzymatic hydrolysis on protein quality and antigenicity of chickpea protein isolate. J. Agr. Food Chem. 47: 3776-3781.
FDA. 1996. Direct food substances affirmed as Generally Recognized As Safe; Enzyme-modified lecithin. Food and Drug Admin., Fed. Reg. 61: 45886-45889.
Fleming, S.E. and Groot Wassink, J.W.D. 1979. Preparation of high fructose syrup from the tubers of the Jerusalem artichoke (Helianthus tuberosus L.). CRC Crit. Rev. Food Sci. Nutr. 12: 1-28.
Fuchs, A. 1993. Production and utilization of inulin: II. Utilization of inulin. In b�0;0;0;0;0;0;0;0;0;0;0;1C;Science and Technology of Fructans,b�0;0;0;0;0;0;0;0;0;0;0;1D; ed. S. Michio and N.J. Chatterton, N.J., pp. 319-352. CRC Press, Boca Raton, Fla.
Gauthier, S.F. and Pouliot, Y. 2003. Functional and biological properties of peptides obtained by enzymatic hydrolysis of whey proteins. J. Dairy Sci. 86(E Suppl.): E78-E87.
Genencor. 2004. Genencor and Health Protection Agency, Porton Down, develop method to inactivate prions. www.genencor.com, Feb. 24.
Godfrey, T. and West, S. 1996. b�0;0;0;0;0;0;0;0;0;0;0;1C;Industrial Enzymology,b�0;0;0;0;0;0;0;0;0;0;0;1D; 2nd ed.Macmillan Press Ltd., London, U.K.
Grassin, C. and Fauquembergue, P. 1996. Fruit juices. In Godfrey and West (1996), pp. 225-264.
Hedges A.R. 1998. Industrial applications of cyclodextrins. Chem. Rev. 98: 2035-2044.
Iametti, S., Rasmussen, P., FrC8kiC&r, H., Ferranti, P., Addeo, P., and Bonomi, F. 2002. Proteolysis of bovine C�0;0;0;0;0;0;0;0;0;0;0;1F;-lactoglobulin during thermal treatment in subdenaturing conditions highlights some structural features of the temperature-modified protein and yields fragments with low immunoreactivity. Eur. J. Biochem. 269(5): 1362.
King et al. 2003. Antibacterial composition for control of gram positive bacteria in food applications. U.S. patent 6,620,446.
Koike, S., Murase, T., and Hase, T. 2003. Oil or fat consumption. U.S. patent application 2003/0054082 A1.
Masui, K., Katsuragi, Y., Tomoko, T., Yasukama, S. 2003. Fat or oil consumption. U.S. patent application 2003/0096867 A1.
Matthews, S.L., Byrne, H., and Hennigan, G.P. 2001. Preparation of a low carbohydrate beer by mashing at high temperature with glucoamylase. J. Inst. Brewing 107(3): 185-194.
Min, S., Mistry, B.S., and Lee, H.O. 2003. Improvement of oxidative and emulsion stability of model salad dressing by glucose oxidase-catalase. J. Food Sci. 68: 1272-1275.
Mokdad et al. 2004. Actual causes of death in the United States, 2000. J. Am. Med. Assn. 291:1238-1245.
Ob�0;0;0;0;0;0;0;0;0;0;0;19;Rourke, T. 1996. Brewing. In Godfrey and West (1996), pp.103-131.
Sato, M., Shimizu, M., Kohori, J., Kase, M., Watanabe, T., and Onozuka, K. 2003. Process for the production of diglycerides. World Patent Application WO 03/060139 A1.
Scheels, N. 2002. Method to bind enzyme to carrier using cationic co-polymers and product produced thereby. World Intellectual Property Organization patent WO 02/083741 A2.
Strauss, K.M., Geller, H.H., and Wagner, B.J. 1979. Preparation of beer with reduced calories. U.S. patent 4,138,499.
WHO. 2004. Fact sheetb�0;0;0;0;0;0;0;0;0;0;0;14;Obesity and overweight. World Health Org., Geneva, Switzerland. www.who.int/hpr/gs.fs.obesity.shtml (accessed March).
